Velocity Constant Of A Reaction At 290K . At 300 k it will be_____, if temperature coefficient. Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. Factors influencing rate of a reaction. When the temperature is raised to 310 k , it will be. The velocity constant of a reaction at 290 k was found to be 3.2 ×. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. When the temperature is raised to 310 k, it will be. When the temperature is raised to 310 k, it will.
from www.bartleby.com
the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. The velocity constant of a reaction at 290 k was found to be 3.2 ×. At 300 k it will be_____, if temperature coefficient. When the temperature is raised to 310 k, it will. When the temperature is raised to 310 k , it will be. When the temperature is raised to 310 k, it will be. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. Factors influencing rate of a reaction. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k.
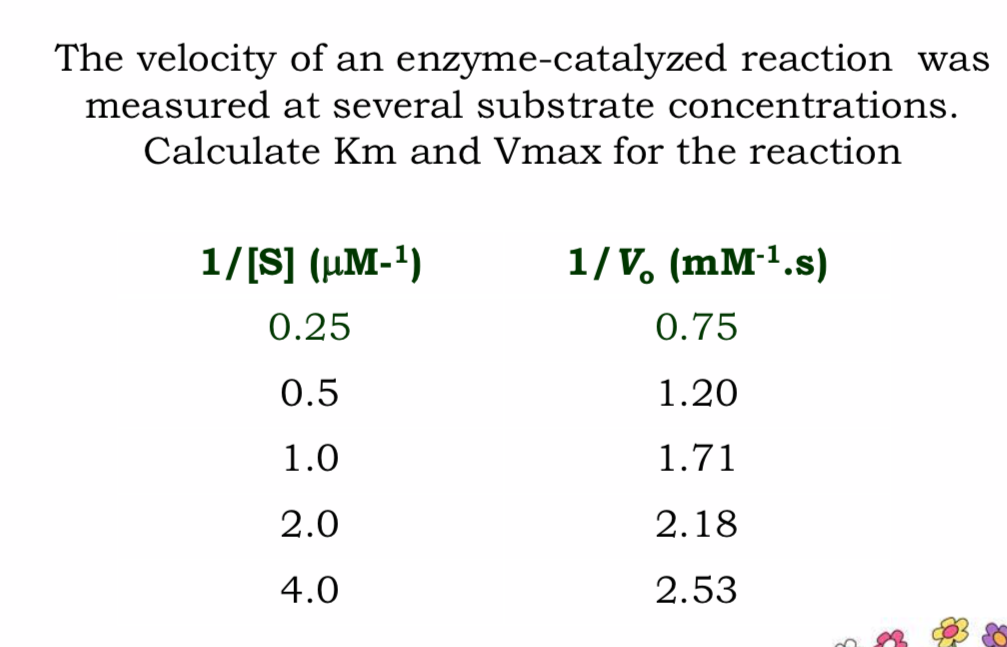
Answered The velocity of an enzymecatalyzed… bartleby
Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k, it will. When the temperature is raised to 310 k, it will. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. Factors influencing rate of a reaction. When the temperature is raised to 310 k, it will be. At 300 k it will be_____, if temperature coefficient. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. When the temperature is raised to 310 k , it will be. The velocity constant of a reaction at 290 k was found to be 3.2 ×.
From the-physics-city.blogspot.com
Physics Constant Velocity Velocity Constant Of A Reaction At 290K At 300 k it will be_____, if temperature coefficient. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. When the temperature is raised to 310 k, it will. When the temperature is raised to 310 k, it will be. Velocity constant of a reaction at 290 k. Velocity Constant Of A Reaction At 290K.
From www.doubtnut.com
The velocity constant of a reaction at 290 K was found to be 3.2 xx 10 Velocity Constant Of A Reaction At 290K At 300 k it will be_____, if temperature coefficient. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. As we know that the velocity constant become double by increasing the temperature by 10∘c so if. Velocity Constant Of A Reaction At 290K.
From www.toppr.com
Velocity constant of a reaction at 290 K was found to be 3.2 × 10^3 Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k, it will. Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. When the temperature is raised to 310 k , it will be. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s −. Velocity Constant Of A Reaction At 290K.
From www.doubtnut.com
IF velocity constant of any reaction [k'] is two times of veloci Velocity Constant Of A Reaction At 290K At 300 k it will be_____, if temperature coefficient. When the temperature is raised to 310 k , it will be. When the temperature is raised to 310 k, it will be. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. The velocity constant of a reaction. Velocity Constant Of A Reaction At 290K.
From www.youtube.com
L05Order of reaction, Units of velocity constant, numerical, Bsc Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k , it will be. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. Factors influencing rate of a reaction. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. Velocity. Velocity Constant Of A Reaction At 290K.
From study.com
Constant Velocity Definition, Equation & Graph Lesson Velocity Constant Of A Reaction At 290K the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. When the temperature is raised to 310 k, it will. When the temperature is raised to 310 k, it will be. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. The velocity constant of a. Velocity Constant Of A Reaction At 290K.
From www.studypool.com
SOLUTION Velocity of reaction examples Studypool Velocity Constant Of A Reaction At 290K Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. When the temperature is raised to 310 k, it will be. When the temperature is raised to 310 k, it will. The velocity constant of a reaction at 290 k was found to be 3.2 ×. the velocity constant of a reaction at 290 k was found. Velocity Constant Of A Reaction At 290K.
From www.youtube.com
The velocity of a particle moving in the positive direction of the X Velocity Constant Of A Reaction At 290K Factors influencing rate of a reaction. When the temperature is raised to 310 k, it will be. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. When the temperature is raised to 310 k, it will. As we know that the velocity constant become double by increasing. Velocity Constant Of A Reaction At 290K.
From www.ausgas.co
constant velocity motion constant velocity transmission Aep22 Velocity Constant Of A Reaction At 290K At 300 k it will be_____, if temperature coefficient. Factors influencing rate of a reaction. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. Velocity constant of a. Velocity Constant Of A Reaction At 290K.
From www.youtube.com
Constant Velocity Overview (Concepts, Variable Isolation, & Graphs Velocity Constant Of A Reaction At 290K Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. When the temperature is raised to 310 k, it will. At 300 k it will be_____, if temperature coefficient. The velocity constant of a reaction at 290 k was found to be 3.2 ×. the velocity constant of a reaction at 290. Velocity Constant Of A Reaction At 290K.
From www.doubtnut.com
Velocity constant of a reactin at 290 K was found to be 3.2 xx 10^(3) Velocity Constant Of A Reaction At 290K Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. Factors influencing rate of a reaction. When. Velocity Constant Of A Reaction At 290K.
From kmbphysics.weebly.com
Unit 1 Kinematics Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k, it will be. At 300 k it will be_____, if temperature coefficient. Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. Velocity. Velocity Constant Of A Reaction At 290K.
From www.youtube.com
Position and Displacement Equation (for Constant Velocity Motion Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k , it will be. Factors influencing rate of a reaction. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. At 300 k it will be_____,. Velocity Constant Of A Reaction At 290K.
From www.doubtnut.com
the velocity constant of the reaction Velocity Constant Of A Reaction At 290K Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. The velocity constant of a reaction at 290 k was found to be 3.2 ×. As we know that the velocity. Velocity Constant Of A Reaction At 290K.
From www.doubtnut.com
The velocity constant of a reaction at 290 K was found to be 3.2 xx 10 Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k , it will be. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. When the temperature is raised to 310 k, it will. At 300 k it will be_____, if temperature coefficient. Velocity constant of a reaction at 290. Velocity Constant Of A Reaction At 290K.
From www.toppr.com
Velocity constant of a reaction at 290 K was found to be 3.2 × 10^3 Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k, it will. The velocity constant of a reaction at 290 k was found to be 3.2 ×. At 300 k it will be_____, if temperature coefficient. Factors influencing rate of a reaction. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s. Velocity Constant Of A Reaction At 290K.
From www.slideserve.com
PPT Kinematics equations for motion with constant acceleration Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k, it will be. Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. At 300 k it will be_____, if temperature coefficient. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. As we know that the velocity constant become double by increasing. Velocity Constant Of A Reaction At 290K.
From calculator.academy
Reaction Velocity Calculator Calculator Academy Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k , it will be. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. The velocity constant of a reaction at 290 k was found to be 3.2 ×. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s −. Velocity Constant Of A Reaction At 290K.
From www.bartleby.com
Answered The velocity of an enzymecatalyzed… bartleby Velocity Constant Of A Reaction At 290K As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. When the temperature is raised to 310 k, it will be. The velocity constant of a reaction at 290 k was found to be 3.2 ×. Factors influencing rate of a reaction. At 300 k it will be_____, if temperature coefficient.. Velocity Constant Of A Reaction At 290K.
From www.slideserve.com
PPT Chapter 14 Chemical Equilibrium PowerPoint Presentation, free Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k, it will. Factors influencing rate of a reaction. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. At 300 k it will be_____, if temperature coefficient. When. Velocity Constant Of A Reaction At 290K.
From brainly.in
what is molecularity and order of reaction? write unit of velocity Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k, it will. When the temperature is raised to 310 k , it will be. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. the velocity constant of a reaction. Velocity Constant Of A Reaction At 290K.
From researchguides.library.vanderbilt.edu
Reaction velocity over time BSCI 1510L Literature and Stats Guide Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k, it will. Factors influencing rate of a reaction. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. At 300 k. Velocity Constant Of A Reaction At 290K.
From www.toppr.com
Two first order reactions proceed at same rate at 298 K when started Velocity Constant Of A Reaction At 290K the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. Factors influencing rate of a reaction. When the temperature is raised to 310 k , it will be. The velocity constant of a reaction at 290 k was found to be 3.2 ×. As we know that the. Velocity Constant Of A Reaction At 290K.
From techdiagrammer.com
The Ultimate Guide to Motion Diagrams Analyzing Velocity Vectors Velocity Constant Of A Reaction At 290K Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. When the temperature is raised to 310 k, it will be. Factors influencing rate of a reaction. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. Velocity constant of a reaction at 290 k was found to be. Velocity Constant Of A Reaction At 290K.
From www.toppr.com
The gaseous reaction A (g) → 2B (g) + (g) obeys first order Velocity Constant Of A Reaction At 290K As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. When the temperature is raised to 310 k, it will. Factors influencing rate of a reaction. When the temperature is raised to 310 k, it will be. At 300 k it will be_____, if temperature coefficient. When the temperature is raised. Velocity Constant Of A Reaction At 290K.
From www.slideserve.com
PPT Motion with Constant Velocity in 1D PowerPoint Presentation, free Velocity Constant Of A Reaction At 290K At 300 k it will be_____, if temperature coefficient. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. Factors influencing rate of a reaction. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. As we know that the velocity constant become double by increasing. Velocity Constant Of A Reaction At 290K.
From www.toppr.com
For a first order reaction velocity constant, k = 10^3s^1 . Two third Velocity Constant Of A Reaction At 290K At 300 k it will be_____, if temperature coefficient. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. The velocity constant of a reaction at 290 k was found to be 3.2 ×. When the. Velocity Constant Of A Reaction At 290K.
From askfilo.com
If a first order reaction completed to 75 in 6 hours the velocity consta.. Velocity Constant Of A Reaction At 290K Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. When the temperature is raised to 310 k, it will be. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. When the temperature is raised to 310 k , it will be. As we know that the velocity constant become. Velocity Constant Of A Reaction At 290K.
From www.slideserve.com
PPT Chapter 2 Enzyme PowerPoint Presentation, free download ID3561286 Velocity Constant Of A Reaction At 290K the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. When the temperature is raised to 310 k , it will be. When the temperature is raised to 310 k, it will be. Velocity constant of a reaction at 290 k was found to be 3.2 × 10. Velocity Constant Of A Reaction At 290K.
From www.youtube.com
CHEMICAL 06 VELOCITY CONSTANT OF REACTION and ARRHENIUS Velocity Constant Of A Reaction At 290K When the temperature is raised to 310 k, it will be. When the temperature is raised to 310 k, it will. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. The velocity constant of a reaction at 290 k was found to be 3.2 ×. Factors influencing. Velocity Constant Of A Reaction At 290K.
From www.youtube.com
Chemical 02 Rate law Velocity constant units Rate Velocity Constant Of A Reaction At 290K Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. As we know that the velocity constant become double by increasing the temperature by 10∘c so if at 290k. the velocity constant of a reaction at 290 k was found. Velocity Constant Of A Reaction At 290K.
From www.youtube.com
Velocity Constant of Second Order Reaction with different initial Velocity Constant Of A Reaction At 290K At 300 k it will be_____, if temperature coefficient. Factors influencing rate of a reaction. When the temperature is raised to 310 k, it will. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. Velocity. Velocity Constant Of A Reaction At 290K.
From www.youtube.com
Velocity and Acceleration Constant Velocity YouTube Velocity Constant Of A Reaction At 290K Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. When the temperature is raised to 310 k, it will be. When the temperature is raised to 310 k , it will be. the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. When the temperature. Velocity Constant Of A Reaction At 290K.
From www.youtube.com
Constant Velocity Graph YouTube Velocity Constant Of A Reaction At 290K Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. When the temperature is raised to 310 k, it will be. Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. The velocity constant of a reaction at 290 k was found to be 3.2 ×. As we know that the. Velocity Constant Of A Reaction At 290K.
From www.slideserve.com
PPT Biochemical Reaction Rate Enzyme PowerPoint Velocity Constant Of A Reaction At 290K the velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3 s − 1. Velocity constant of a reaction at 290 k was found to be 3.2 × 10 − 3. Velocity constant of a reaction at 290 k was found to 3.2×10−3sec−1. The velocity constant of a reaction at 290 k. Velocity Constant Of A Reaction At 290K.