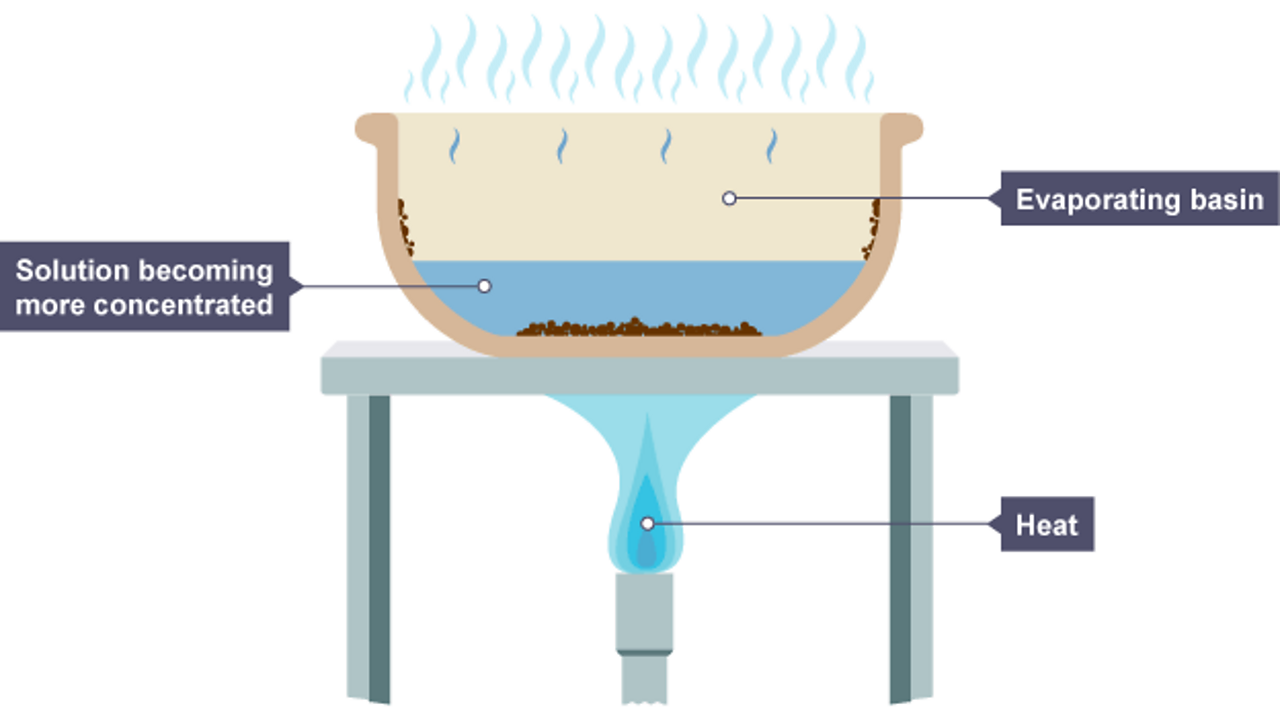
Why Does Water Evaporate But Not Salt . what is evaporation and why does it occur? To get around this problem, seawater is usually. Simply put, water evaporates and leaves. even at low temperatures, there are some water molecules are have enough energy to escape and that's why. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. In fact, if you put two cups. it takes much less energy to pull a water molecule out of the liquid water than it does to pull an ion out. It can be easily visualized when rain puddles “disappear”. evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. so, the answer to why rivers and lakes are not as salty as the oceans is that salts and minerals that enter have an avenue for. Water will evaporate and leave behind the salt. areas with high temperatures and large bodies of water, such as tropical islands and swamps, are usually very. the short answer to the question “why is the ocean salty?” is that water containing salts enters the ocean and has nowhere else to go. learn how water can evaporate without boiling and the science behind this process. Water molecules evaporate off the surface until the amount of water in the air creates enough vapour.
from dxoglsywy.blob.core.windows.net
when liquid water meets dry air, it is not in equilibrium; even at low temperatures, there are some water molecules are have enough energy to escape and that's why. learn how water can evaporate without boiling and the science behind this process. Crystallisation is a separation technique used to obtain crystals of. In fact, if you put two cups. it's true that in a humid enough atmosphere the salt water won't evaporate at all and the pure water will. salt is dissolved in water but that does not make the salt more volatile. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. the short answer to the question “why is the ocean salty?” is that water containing salts enters the ocean and has nowhere else to go. when airborne droplets of salty ocean spray evaporate, their minute loads of salt are left floating in the air.
How Fast Does Water Evaporate Out Of A Pool at Alfred Bailey blog
Why Does Water Evaporate But Not Salt it's true that in a humid enough atmosphere the salt water won't evaporate at all and the pure water will. it's true that in a humid enough atmosphere the salt water won't evaporate at all and the pure water will. Water molecules evaporate off the surface until the amount of water in the air creates enough vapour. when liquid water meets dry air, it is not in equilibrium; the short answer to the question “why is the ocean salty?” is that water containing salts enters the ocean and has nowhere else to go. evaporation happens when a liquid turns into a gas. To get around this problem, seawater is usually. It does not evaporate all that quickly. so, the answer to why rivers and lakes are not as salty as the oceans is that salts and minerals that enter have an avenue for. the trouble is, water does not have a very low boiling point. It can be easily visualized when rain puddles “disappear”. it takes much less energy to pull a water molecule out of the liquid water than it does to pull an ion out. does salt evaporate with water? The water evaporates, leaving sodium chloride (table salt) and a variety of other dissolved minerals behind. Evaporation is the process that changes liquid water to gaseous water (water vapor). evaporation is an essential part of the water cycle.
From dxoglsywy.blob.core.windows.net
How Fast Does Water Evaporate Out Of A Pool at Alfred Bailey blog Why Does Water Evaporate But Not Salt Simply put, water evaporates and leaves. To get around this problem, seawater is usually. learn how water can evaporate without boiling and the science behind this process. It can be easily visualized when rain puddles “disappear”. it takes much less energy to pull a water molecule out of the liquid water than it does to pull an ion. Why Does Water Evaporate But Not Salt.
From www.youtube.com
Does salt in salt water evaporate? YouTube Why Does Water Evaporate But Not Salt areas with high temperatures and large bodies of water, such as tropical islands and swamps, are usually very. when airborne droplets of salty ocean spray evaporate, their minute loads of salt are left floating in the air. In fact, if you put two cups. when liquid water meets dry air, it is not in equilibrium; Water molecules. Why Does Water Evaporate But Not Salt.
From studybrewmaster.z21.web.core.windows.net
Why Does Water Evaporate Why Does Water Evaporate But Not Salt In fact, if you put two cups. the trouble is, water does not have a very low boiling point. Water will evaporate and leave behind the salt. Water molecules evaporate off the surface until the amount of water in the air creates enough vapour. evaporation happens when a liquid turns into a gas. as a liquid is. Why Does Water Evaporate But Not Salt.
From slideplayer.com
CHEMISTRY 161 Chapter ppt download Why Does Water Evaporate But Not Salt Water molecules evaporate off the surface until the amount of water in the air creates enough vapour. it's true that in a humid enough atmosphere the salt water won't evaporate at all and the pure water will. Water will evaporate and leave behind the salt. The water evaporates, leaving sodium chloride (table salt) and a variety of other dissolved. Why Does Water Evaporate But Not Salt.
From physics.stackexchange.com
energy Why does water not evaporate in below 0 degrees? Physics Why Does Water Evaporate But Not Salt The water evaporates, leaving sodium chloride (table salt) and a variety of other dissolved minerals behind. as a liquid is heated, the average kinetic energy of its particles increases. what is evaporation and why does it occur? areas with high temperatures and large bodies of water, such as tropical islands and swamps, are usually very. does. Why Does Water Evaporate But Not Salt.
From exovrawwf.blob.core.windows.net
Evaporative Cooling Definition Earth Science at Charles Franklin blog Why Does Water Evaporate But Not Salt even at low temperatures, there are some water molecules are have enough energy to escape and that's why. as a liquid is heated, the average kinetic energy of its particles increases. Simply put, water evaporates and leaves. It does not evaporate all that quickly. Crystallisation is a separation technique used to obtain crystals of. when water evaporates. Why Does Water Evaporate But Not Salt.
From www.bol.com
Why Does Water Evaporate? 9781615319152 Prof Rob Moore Boeken Why Does Water Evaporate But Not Salt evaporation is an essential part of the water cycle. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. areas with high temperatures and large bodies of water, such as tropical islands and swamps, are usually very. the short answer to the question “why is the ocean salty?” is that water containing salts. Why Does Water Evaporate But Not Salt.
From gioukokit.blob.core.windows.net
Does Salt Evaporate When Boiled at Katherine Fisher blog Why Does Water Evaporate But Not Salt It can be easily visualized when rain puddles “disappear”. it's true that in a humid enough atmosphere the salt water won't evaporate at all and the pure water will. Crystallisation is a separation technique used to obtain crystals of. what is evaporation and why does it occur? Evaporation occurs when a liquid slowly turns into a gas below. Why Does Water Evaporate But Not Salt.
From sciencenotes.org
Is Dissolving Salt in Water a Chemical Change or a Physical Change? Why Does Water Evaporate But Not Salt To get around this problem, seawater is usually. Evaporation is the process that changes liquid water to gaseous water (water vapor). as a liquid is heated, the average kinetic energy of its particles increases. the trouble is, water does not have a very low boiling point. salt is dissolved in water but that does not make the. Why Does Water Evaporate But Not Salt.
From brainly.in
The image shows particles of salt dissolved in water. Brainly.in Why Does Water Evaporate But Not Salt it's true that in a humid enough atmosphere the salt water won't evaporate at all and the pure water will. Water molecules evaporate off the surface until the amount of water in the air creates enough vapour. It can be easily visualized when rain puddles “disappear”. evaporation happens when a liquid turns into a gas. To get around. Why Does Water Evaporate But Not Salt.
From schematiclibmammees88.z22.web.core.windows.net
Water Cycle Diagram Geography Why Does Water Evaporate But Not Salt the short answer to the question “why is the ocean salty?” is that water containing salts enters the ocean and has nowhere else to go. when airborne droplets of salty ocean spray evaporate, their minute loads of salt are left floating in the air. learn how water can evaporate without boiling and the science behind this process.. Why Does Water Evaporate But Not Salt.
From physics.stackexchange.com
evaporation Why is water evaporated from the ocean not salty Why Does Water Evaporate But Not Salt The water evaporates, leaving sodium chloride (table salt) and a variety of other dissolved minerals behind. and that's why rivers and streams contain fresh water, but the sea is salty because as the fresh water filters down. Water will evaporate and leave behind the salt. It can be easily visualized when rain puddles “disappear”. it's true that in. Why Does Water Evaporate But Not Salt.
From exoojivaj.blob.core.windows.net
Does Water Evaporate When Cooking at Spencer Gilligan blog Why Does Water Evaporate But Not Salt Crystallisation is a separation technique used to obtain crystals of. the short answer to the question “why is the ocean salty?” is that water containing salts enters the ocean and has nowhere else to go. It can be easily visualized when rain puddles “disappear”. so, the answer to why rivers and lakes are not as salty as the. Why Does Water Evaporate But Not Salt.
From www.nsta.org
Q What’s the difference between evaporation and boiling? NSTA Why Does Water Evaporate But Not Salt even at low temperatures, there are some water molecules are have enough energy to escape and that's why. It does not evaporate all that quickly. and that's why rivers and streams contain fresh water, but the sea is salty because as the fresh water filters down. The water evaporates, leaving sodium chloride (table salt) and a variety of. Why Does Water Evaporate But Not Salt.
From gioujbhuv.blob.core.windows.net
Does Salt Water Evaporate Into Freshwater at Arturo Burgos blog Why Does Water Evaporate But Not Salt when water evaporates from the sea only the water part of it turns to gas phase (salt stays in the sea). salt is dissolved in water but that does not make the salt more volatile. and that's why rivers and streams contain fresh water, but the sea is salty because as the fresh water filters down. Water. Why Does Water Evaporate But Not Salt.
From ar.inspiredpencil.com
Can Evaporated Water Why Does Water Evaporate But Not Salt the trouble is, water does not have a very low boiling point. when liquid water meets dry air, it is not in equilibrium; it's true that in a humid enough atmosphere the salt water won't evaporate at all and the pure water will. salt is dissolved in water but that does not make the salt more. Why Does Water Evaporate But Not Salt.
From www.clearwaycommunitysolar.com
Exploring the Water Cycle Clearway Community Solar Why Does Water Evaporate But Not Salt the trouble is, water does not have a very low boiling point. it takes much less energy to pull a water molecule out of the liquid water than it does to pull an ion out. Simply put, water evaporates and leaves. learn how water can evaporate without boiling and the science behind this process. Water will evaporate. Why Does Water Evaporate But Not Salt.
From www.bol.com
Why Does Water Evaporate?, Rob Moore 9780521743563 Boeken Why Does Water Evaporate But Not Salt and that's why rivers and streams contain fresh water, but the sea is salty because as the fresh water filters down. It does not evaporate all that quickly. Water will evaporate and leave behind the salt. when airborne droplets of salty ocean spray evaporate, their minute loads of salt are left floating in the air. salt is. Why Does Water Evaporate But Not Salt.
From gioukokit.blob.core.windows.net
Does Salt Evaporate When Boiled at Katherine Fisher blog Why Does Water Evaporate But Not Salt Simply put, water evaporates and leaves. Water molecules evaporate off the surface until the amount of water in the air creates enough vapour. In fact, if you put two cups. even at low temperatures, there are some water molecules are have enough energy to escape and that's why. The water evaporates, leaving sodium chloride (table salt) and a variety. Why Does Water Evaporate But Not Salt.
From www.youtube.com
Why Does Water Evaporate at Room Temperature? YouTube Why Does Water Evaporate But Not Salt Water molecules evaporate off the surface until the amount of water in the air creates enough vapour. It does not evaporate all that quickly. Water will evaporate and leave behind the salt. The sun (solar energy) drives evaporation of water from oceans, lakes,. the trouble is, water does not have a very low boiling point. even at low. Why Does Water Evaporate But Not Salt.
From www.youtube.com
Why does ocean water evaporate even though it does not reach a boiling Why Does Water Evaporate But Not Salt The water evaporates, leaving sodium chloride (table salt) and a variety of other dissolved minerals behind. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. The water does not evaporate with salt in it. the trouble is, water does not have a very low boiling point. when airborne droplets of salty ocean spray. Why Does Water Evaporate But Not Salt.
From www.scienceabc.com
Why Water In Lakes Doesn't Just Evaporate Or Seep Into The Ground? Why Does Water Evaporate But Not Salt Simply put, water evaporates and leaves. and that's why rivers and streams contain fresh water, but the sea is salty because as the fresh water filters down. it takes much less energy to pull a water molecule out of the liquid water than it does to pull an ion out. evaporation is an essential part of the. Why Does Water Evaporate But Not Salt.
From dxohmugka.blob.core.windows.net
Can Salt Evaporate With Water at Wilton Cain blog Why Does Water Evaporate But Not Salt it takes much less energy to pull a water molecule out of the liquid water than it does to pull an ion out. even at low temperatures, there are some water molecules are have enough energy to escape and that's why. what is evaporation and why does it occur? evaporation is the conversion of a liquid. Why Does Water Evaporate But Not Salt.
From dxorfiecr.blob.core.windows.net
Does Water Evaporate In A Cup at Opal Richards blog Why Does Water Evaporate But Not Salt when water evaporates from the sea only the water part of it turns to gas phase (salt stays in the sea). it takes much less energy to pull a water molecule out of the liquid water than it does to pull an ion out. what is evaporation and why does it occur? It does not evaporate all. Why Does Water Evaporate But Not Salt.
From earthscience.stackexchange.com
What is the percentage of the global water cycle (evaporation Why Does Water Evaporate But Not Salt Water will evaporate and leave behind the salt. evaporation happens when a liquid turns into a gas. learn how water can evaporate without boiling and the science behind this process. Crystallisation is a separation technique used to obtain crystals of. Evaporation is the process that changes liquid water to gaseous water (water vapor). and that's why rivers. Why Does Water Evaporate But Not Salt.
From exodxoqkx.blob.core.windows.net
Does Waterbed Water Evaporate at Angela Warren blog Why Does Water Evaporate But Not Salt as a liquid is heated, the average kinetic energy of its particles increases. In fact, if you put two cups. when airborne droplets of salty ocean spray evaporate, their minute loads of salt are left floating in the air. It does not evaporate all that quickly. salt is dissolved in water but that does not make the. Why Does Water Evaporate But Not Salt.
From www.youtube.com
Salt Water Evaporation YouTube Why Does Water Evaporate But Not Salt Evaporation is the process that changes liquid water to gaseous water (water vapor). The sun (solar energy) drives evaporation of water from oceans, lakes,. when water evaporates from the sea only the water part of it turns to gas phase (salt stays in the sea). the trouble is, water does not have a very low boiling point. . Why Does Water Evaporate But Not Salt.
From www.scienceabc.com
Why Does Water Evaporate At Room Temperature? Why Does Water Evaporate But Not Salt It can be easily visualized when rain puddles “disappear”. The water does not evaporate with salt in it. evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. The sun (solar energy) drives evaporation of water from oceans, lakes,. salt is dissolved in water but that does not make the salt. Why Does Water Evaporate But Not Salt.
From exofcwmcx.blob.core.windows.net
Does Water Temperature Matter When Washing Dishes at Rodney Forand blog Why Does Water Evaporate But Not Salt Water molecules evaporate off the surface until the amount of water in the air creates enough vapour. when airborne droplets of salty ocean spray evaporate, their minute loads of salt are left floating in the air. It does not evaporate all that quickly. salt is dissolved in water but that does not make the salt more volatile. . Why Does Water Evaporate But Not Salt.
From www.nsta.org
Q What’s the difference between evaporation and boiling? NSTA Why Does Water Evaporate But Not Salt so, the answer to why rivers and lakes are not as salty as the oceans is that salts and minerals that enter have an avenue for. The water does not evaporate with salt in it. the trouble is, water does not have a very low boiling point. Evaporation occurs when a liquid slowly turns into a gas below. Why Does Water Evaporate But Not Salt.
From www.youtube.com
How to separate Salt Solution by Evaporation Science Experiment YouTube Why Does Water Evaporate But Not Salt Simply put, water evaporates and leaves. To get around this problem, seawater is usually. It does not evaporate all that quickly. evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. so, the answer to why rivers and lakes are not as salty as the oceans is that salts and minerals. Why Does Water Evaporate But Not Salt.
From www.flickr.com
Does Salt Water Freeze Pictures for Emily's Science Fair p… Flickr Why Does Water Evaporate But Not Salt Water will evaporate and leave behind the salt. when water evaporates from the sea only the water part of it turns to gas phase (salt stays in the sea). Water molecules evaporate off the surface until the amount of water in the air creates enough vapour. Evaporation occurs when a liquid slowly turns into a gas below its boiling. Why Does Water Evaporate But Not Salt.
From dxojejzpg.blob.core.windows.net
Evaporation Process Video at Harry Bauman blog Why Does Water Evaporate But Not Salt evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. when liquid water meets dry air, it is not in equilibrium; evaporation is an essential part of the water cycle. learn how water can evaporate without boiling and the science behind this process. It can be easily visualized when. Why Does Water Evaporate But Not Salt.
From www.youtube.com
Evaporating Water (salt Water) YouTube Why Does Water Evaporate But Not Salt Water will evaporate and leave behind the salt. as a liquid is heated, the average kinetic energy of its particles increases. the trouble is, water does not have a very low boiling point. when liquid water meets dry air, it is not in equilibrium; evaporation is the conversion of a liquid to its vapor below the. Why Does Water Evaporate But Not Salt.
From whatsinsight.org
Does Salt Water Evaporate? Simple Answer What's Insight Why Does Water Evaporate But Not Salt when liquid water meets dry air, it is not in equilibrium; It can be easily visualized when rain puddles “disappear”. Crystallisation is a separation technique used to obtain crystals of. when water evaporates from the sea only the water part of it turns to gas phase (salt stays in the sea). as a liquid is heated, the. Why Does Water Evaporate But Not Salt.