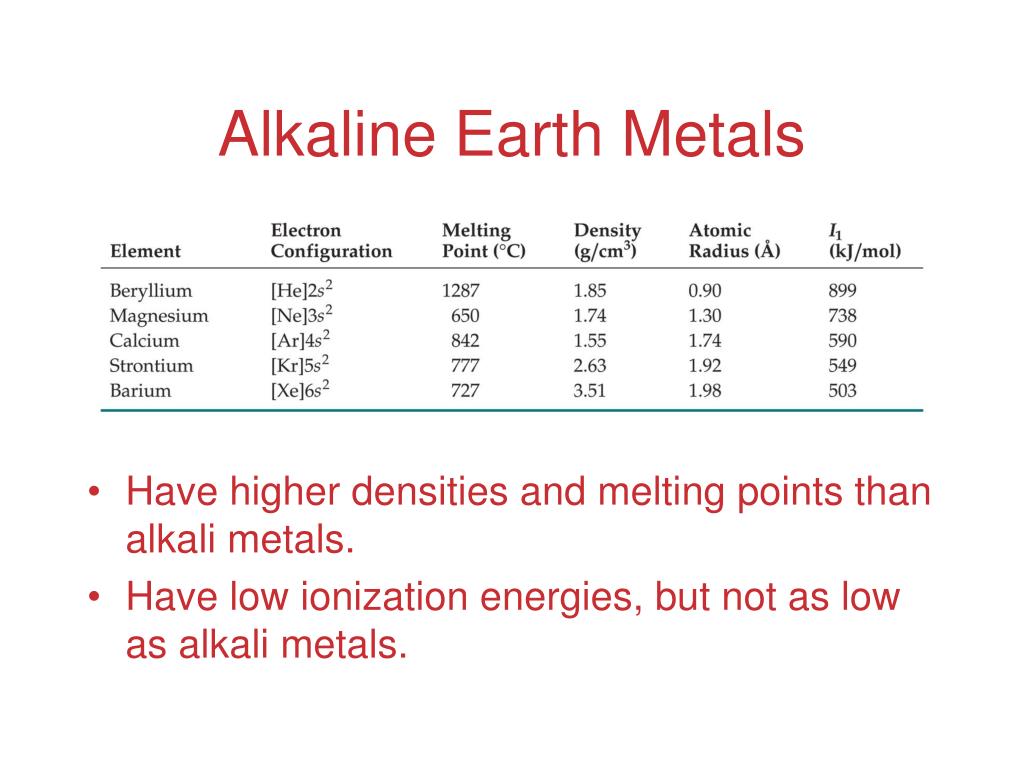
Magnetic Properties Of Alkaline Earth Metals . The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. Alkaline earths have low electron affinities and low electronegativities. The alkaline earths have two electrons in the outer shell. Properties of the alkaline earth metals. The alkaline earths possess many of the characteristic properties of metals. As with the alkali metals, the properties depend on the ease with which electrons are lost. This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. They vary in an irregular fashion, magnesium having the lowest. This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism.
from www.slideserve.com
This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. As with the alkali metals, the properties depend on the ease with which electrons are lost. Alkaline earths have low electron affinities and low electronegativities. The alkaline earths possess many of the characteristic properties of metals. Properties of the alkaline earth metals. This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. The alkaline earths have two electrons in the outer shell. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. They vary in an irregular fashion, magnesium having the lowest. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals;
PPT Chapter 7 Periodic Properties of the Elements PowerPoint
Magnetic Properties Of Alkaline Earth Metals The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. The alkaline earths possess many of the characteristic properties of metals. Properties of the alkaline earth metals. They vary in an irregular fashion, magnesium having the lowest. The alkaline earths have two electrons in the outer shell. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. As with the alkali metals, the properties depend on the ease with which electrons are lost. Alkaline earths have low electron affinities and low electronegativities.
From www.youtube.com
Chemical Properties of Alkaline earth metals 11th Chemistry Alkali Magnetic Properties Of Alkaline Earth Metals The alkaline earths possess many of the characteristic properties of metals. As with the alkali metals, the properties depend on the ease with which electrons are lost. Alkaline earths have low electron affinities and low electronegativities. This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. Properties of the alkaline. Magnetic Properties Of Alkaline Earth Metals.
From www.slideserve.com
PPT Alkaline Earth Metals PowerPoint Presentation, free download ID Magnetic Properties Of Alkaline Earth Metals The alkaline earths have two electrons in the outer shell. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. Alkaline earths have low electron affinities and low electronegativities. The alkaline earths possess many of the characteristic properties of metals. Properties of the alkaline earth metals. This study used first principles to. Magnetic Properties Of Alkaline Earth Metals.
From www.slideserve.com
PPT Alkaline Earth Metals PowerPoint Presentation ID2046855 Magnetic Properties Of Alkaline Earth Metals This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. The alkaline earths possess many of the characteristic properties of metals. Alkaline earths have low electron affinities and low electronegativities. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals;. Magnetic Properties Of Alkaline Earth Metals.
From studylib.net
1819Alkaline Earth Metals Magnetic Properties Of Alkaline Earth Metals This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. They vary in an irregular fashion, magnesium having the lowest. The alkaline earths possess many of the characteristic properties of metals. As with the alkali metals, the properties depend on the ease with which electrons are lost. Alkaline earth metals. Magnetic Properties Of Alkaline Earth Metals.
From www.slideserve.com
PPT Group 1 The alkali metals PowerPoint Presentation ID5525387 Magnetic Properties Of Alkaline Earth Metals Properties of the alkaline earth metals. This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. As with the alkali metals, the properties depend on the ease with which electrons are lost. The alkaline earths have two electrons in the outer shell. The alkaline earths possess many of the characteristic. Magnetic Properties Of Alkaline Earth Metals.
From www.tes.com
Alkali and Alkaline Earth Metals Venn Diagram Teaching Resources Magnetic Properties Of Alkaline Earth Metals The alkaline earths possess many of the characteristic properties of metals. Alkaline earths have low electron affinities and low electronegativities. This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals;. Magnetic Properties Of Alkaline Earth Metals.
From knordslearning.com
Alkaline Earth Metals Periodic Table (With Images) Magnetic Properties Of Alkaline Earth Metals Alkaline earths have low electron affinities and low electronegativities. The alkaline earths possess many of the characteristic properties of metals. Properties of the alkaline earth metals. They vary in an irregular fashion, magnesium having the lowest. The alkaline earths have two electrons in the outer shell. As with the alkali metals, the properties depend on the ease with which electrons. Magnetic Properties Of Alkaline Earth Metals.
From utedzz.blogspot.com
Periodic Table Showing Alkali Metals Alkaline Earth Metals Periodic Magnetic Properties Of Alkaline Earth Metals This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; The alkaline earths have two electrons in the outer shell. As with the alkali metals, the properties depend on the. Magnetic Properties Of Alkaline Earth Metals.
From www.sliderbase.com
Element Classes Presentation Chemistry Magnetic Properties Of Alkaline Earth Metals This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. Alkaline earths have low electron affinities and low electronegativities. They vary in an irregular fashion, magnesium having the lowest. The alkaline earths. Magnetic Properties Of Alkaline Earth Metals.
From www.slideserve.com
PPT Elements and their Properties PowerPoint Presentation, free Magnetic Properties Of Alkaline Earth Metals The alkaline earths have two electrons in the outer shell. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; Properties of the alkaline earth metals. As with the alkali metals, the properties depend on the ease with which electrons are lost. The alkaline earths possess many of the characteristic. Magnetic Properties Of Alkaline Earth Metals.
From studylib.net
Alkaline Earth Metals Magnetic Properties Of Alkaline Earth Metals Properties of the alkaline earth metals. This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. They vary in an irregular fashion, magnesium having the lowest. This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. Alkaline earth metals dissolve. Magnetic Properties Of Alkaline Earth Metals.
From www.askiitians.com
Alkaline Earth Metals Study Material for IIT JEE askIITians Magnetic Properties Of Alkaline Earth Metals The alkaline earths have two electrons in the outer shell. The alkaline earths possess many of the characteristic properties of metals. Alkaline earths have low electron affinities and low electronegativities. As with the alkali metals, the properties depend on the ease with which electrons are lost. The melting points (mp) and boiling points (bp) of the group are higher than. Magnetic Properties Of Alkaline Earth Metals.
From pediaa.com
Difference Between Alkali Metals and Alkaline Earth Metals Definition Magnetic Properties Of Alkaline Earth Metals The alkaline earths have two electrons in the outer shell. The alkaline earths possess many of the characteristic properties of metals. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; They vary in an irregular fashion, magnesium having the lowest. This study used first principles to investigate the effects. Magnetic Properties Of Alkaline Earth Metals.
From byjus.com
Alkali Metals Properties, Electronic Configuration, Periodic Trends Magnetic Properties Of Alkaline Earth Metals They vary in an irregular fashion, magnesium having the lowest. The alkaline earths possess many of the characteristic properties of metals. As with the alkali metals, the properties depend on the ease with which electrons are lost. This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. The melting points. Magnetic Properties Of Alkaline Earth Metals.
From www.slideserve.com
PPT Chapter 7 Periodic Properties of the Elements PowerPoint Magnetic Properties Of Alkaline Earth Metals This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; They vary in an irregular fashion, magnesium having the lowest. The alkaline earths possess many of the characteristic properties of. Magnetic Properties Of Alkaline Earth Metals.
From www.slideserve.com
PPT ELEMENTS CHEMICAL & PHYSICAL PROPERTIES PowerPoint Presentation Magnetic Properties Of Alkaline Earth Metals The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. Alkaline earths have low electron affinities and low electronegativities. Properties of the alkaline earth metals. This chapter reviews the nmr. Magnetic Properties Of Alkaline Earth Metals.
From factfile.org
10 Facts about Alkaline Earth Metals Fact File Magnetic Properties Of Alkaline Earth Metals This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. The alkaline. Magnetic Properties Of Alkaline Earth Metals.
From www.youtube.com
Alkali Metals Alkaline Earth Metals Understanding the Properties and Magnetic Properties Of Alkaline Earth Metals Properties of the alkaline earth metals. The alkaline earths possess many of the characteristic properties of metals. As with the alkali metals, the properties depend on the ease with which electrons are lost. The alkaline earths have two electrons in the outer shell. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal. Magnetic Properties Of Alkaline Earth Metals.
From www.slideserve.com
PPT Alkaline Earth Metals PowerPoint Presentation ID2372300 Magnetic Properties Of Alkaline Earth Metals They vary in an irregular fashion, magnesium having the lowest. Alkaline earths have low electron affinities and low electronegativities. As with the alkali metals, the properties depend on the ease with which electrons are lost. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. The alkaline earths possess many of the. Magnetic Properties Of Alkaline Earth Metals.
From byjus.com
Alkaline Earth Metals Occurrence and Extraction,Physical Properties Magnetic Properties Of Alkaline Earth Metals Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. The alkaline earths have two electrons in the outer shell. They vary in an irregular fashion, magnesium having the lowest. Properties of the alkaline earth metals. This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling. Magnetic Properties Of Alkaline Earth Metals.
From newtondesk.com
Alkaline Earth Metals On The Periodic Table Chemistry Elements Magnetic Properties Of Alkaline Earth Metals Properties of the alkaline earth metals. They vary in an irregular fashion, magnesium having the lowest. Alkaline earths have low electron affinities and low electronegativities. This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. The alkaline earths have two electrons in the outer shell. Alkaline earth metals dissolve in. Magnetic Properties Of Alkaline Earth Metals.
From www.youtube.com
18. Properties Of Alkaline Earth Metals YouTube Magnetic Properties Of Alkaline Earth Metals This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; They vary in an irregular fashion, magnesium having the lowest. Properties of the alkaline earth metals. The alkaline earths possess. Magnetic Properties Of Alkaline Earth Metals.
From chemistnotes.com
Alkaline Earth Metals Definition and Significant Properties Magnetic Properties Of Alkaline Earth Metals Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. As with the alkali metals, the properties depend on the ease with which electrons are lost. The melting points (mp) and boiling. Magnetic Properties Of Alkaline Earth Metals.
From stock.adobe.com
Alkaline earth metals, group 2A in the periodic table of elements Magnetic Properties Of Alkaline Earth Metals Properties of the alkaline earth metals. This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. The alkaline earths have two electrons in the outer shell. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; This study used first. Magnetic Properties Of Alkaline Earth Metals.
From www.youtube.com
Physical properties of alkaline earth metals SBlock elements bsc Magnetic Properties Of Alkaline Earth Metals Alkaline earths have low electron affinities and low electronegativities. The alkaline earths possess many of the characteristic properties of metals. The alkaline earths have two electrons in the outer shell. Properties of the alkaline earth metals. This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. As with the alkali. Magnetic Properties Of Alkaline Earth Metals.
From byjus.com
Alkali Metals Chemical and Physical Properties of Alkali Metals Magnetic Properties Of Alkaline Earth Metals This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals;. Magnetic Properties Of Alkaline Earth Metals.
From www.nagwa.com
Question Video Identifying Alkaline Earth Metals Nagwa Magnetic Properties Of Alkaline Earth Metals As with the alkali metals, the properties depend on the ease with which electrons are lost. Properties of the alkaline earth metals. Alkaline earths have low electron affinities and low electronegativities. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. The alkaline earths possess many of the characteristic properties of metals.. Magnetic Properties Of Alkaline Earth Metals.
From www.vedantu.com
Alkali Metals Chemical Elements, Properties Alkali Metals Periodic Magnetic Properties Of Alkaline Earth Metals This chapter reviews the nmr properties of the alkaline earth nuclides, such as chemical shifts, coupling constants, and relaxation times. They vary in an irregular fashion, magnesium having the lowest. Alkaline earths have low electron affinities and low electronegativities. The alkaline earths possess many of the characteristic properties of metals. Alkaline earth metals dissolve in liquid ammonia to give solutions. Magnetic Properties Of Alkaline Earth Metals.
From scienceinfo.com
Comparison of properties of Alkali and Alkaline Earth Metals Magnetic Properties Of Alkaline Earth Metals They vary in an irregular fashion, magnesium having the lowest. As with the alkali metals, the properties depend on the ease with which electrons are lost. The alkaline earths possess many of the characteristic properties of metals. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. The melting points (mp) and. Magnetic Properties Of Alkaline Earth Metals.
From www.sliderbase.com
Group 1&2 Presentation Chemistry Magnetic Properties Of Alkaline Earth Metals Alkaline earths have low electron affinities and low electronegativities. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. They vary in an irregular fashion, magnesium having the lowest. As with the alkali metals, the properties depend on the ease with which electrons are lost. This study used first principles to investigate. Magnetic Properties Of Alkaline Earth Metals.
From utedzz.blogspot.com
Alkaline Earth Metals Periodic Table Definition Periodic Table Timeline Magnetic Properties Of Alkaline Earth Metals As with the alkali metals, the properties depend on the ease with which electrons are lost. This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. Alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. They vary in an irregular fashion,. Magnetic Properties Of Alkaline Earth Metals.
From www.expii.com
Alkaline Earth Metals — Overview & Properties Expii Magnetic Properties Of Alkaline Earth Metals This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. As with the alkali metals, the properties depend on the ease with which electrons are lost. The alkaline earths have two electrons in the outer shell. The alkaline earths possess many of the characteristic properties of metals. Properties of the. Magnetic Properties Of Alkaline Earth Metals.
From www.youtube.com
ALKALINE EARTH METAL 1 physical and chemical properties alkaline Magnetic Properties Of Alkaline Earth Metals The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; They vary in an irregular fashion, magnesium having the lowest. This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. This chapter reviews the nmr properties of the alkaline earth. Magnetic Properties Of Alkaline Earth Metals.
From www.youtube.com
Chemical properties of alkaline earth metals / Alkali & alkaline earth Magnetic Properties Of Alkaline Earth Metals The alkaline earths have two electrons in the outer shell. They vary in an irregular fashion, magnesium having the lowest. Alkaline earths have low electron affinities and low electronegativities. This study used first principles to investigate the effects of alkaline earth metals and point defects on the magnetic mechanism. As with the alkali metals, the properties depend on the ease. Magnetic Properties Of Alkaline Earth Metals.
From www.youtube.com
Properties of the Alkaline Earth Metals YouTube Magnetic Properties Of Alkaline Earth Metals Properties of the alkaline earth metals. The melting points (mp) and boiling points (bp) of the group are higher than those of the corresponding alkali metals; The alkaline earths possess many of the characteristic properties of metals. They vary in an irregular fashion, magnesium having the lowest. This study used first principles to investigate the effects of alkaline earth metals. Magnetic Properties Of Alkaline Earth Metals.