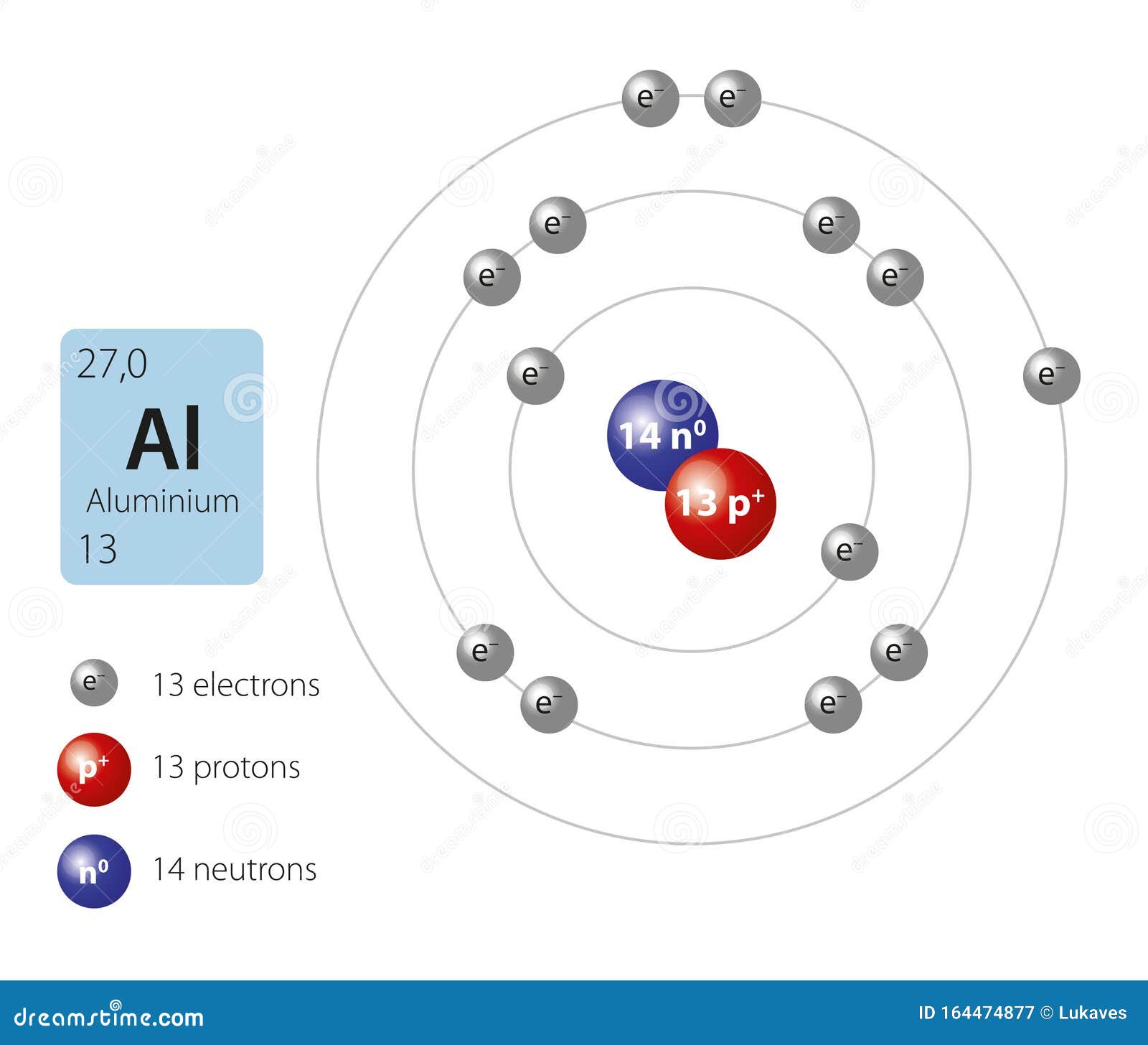
An Aluminum Atom Has A Mass Of 4.48 . Use this information to answer. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer the. Given that the mass of one aluminum. An aluminum atom has a mass of 4. Use this information to answer. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Here’s how to approach this question. 4 8 x 1 0. Calculate the mass of 1 mole of aluminum atoms using.
from rokanggun.blogspot.com
An aluminum atom has a mass of 4. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer. Calculate the mass of 1 mole of aluminum atoms using. Use this information to answer the. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. 4 8 x 1 0. Use this information to answer. Given that the mass of one aluminum. Here’s how to approach this question.
18+ Aluminum Atome
An Aluminum Atom Has A Mass Of 4.48 Calculate the mass of 1 mole of aluminum atoms using. 4 8 x 1 0. the atomic mass of aluminum is approximately 26.98 grams per mole. Here’s how to approach this question. An aluminum atom has a mass of 4. Given that the mass of one aluminum. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer the. Use this information to answer. Calculate the mass of 1 mole of aluminum atoms using. Use this information to answer.
From www.chegg.com
Solved An aluminum atom has a mass of 4.48 * 10^23 grams An Aluminum Atom Has A Mass Of 4.48 Use this information to answer. Use this information to answer. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. the atomic mass of aluminum is approximately 26.98 grams per mole. 4 8 x 1 0. Here’s how to approach this question. An aluminum atom has a mass of 4. Use this. An Aluminum Atom Has A Mass Of 4.48.
From www.chegg.com
Solved 23 An aluminum atom has a mass of 4.48 x 10 g and a An Aluminum Atom Has A Mass Of 4.48 Use this information to answer. Calculate the mass of 1 mole of aluminum atoms using. 4 8 x 1 0. the atomic mass of aluminum is approximately 26.98 grams per mole. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Here’s how to approach this question. Use this information to answer.. An Aluminum Atom Has A Mass Of 4.48.
From www.sciencephoto.com
Aluminium, atomic structure Stock Image C013/1526 Science Photo An Aluminum Atom Has A Mass Of 4.48 The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. 4 8 x 1 0. the atomic mass of aluminum is approximately 26.98 grams per mole. An aluminum atom has a mass of 4. Use this information to answer. Given that the mass of one aluminum. Here’s how to approach this question.. An Aluminum Atom Has A Mass Of 4.48.
From www.museoinclusivo.com
Exploring Aluminum Atomic Mass Properties, Uses, and Significance An Aluminum Atom Has A Mass Of 4.48 Use this information to answer. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Here’s how to approach this question. Given that the mass of one aluminum. Use this information to answer the. Calculate the mass of 1 mole of aluminum atoms using. Use this information to answer. the atomic mass. An Aluminum Atom Has A Mass Of 4.48.
From www.sciencephoto.com
Aluminum, atomic structure Stock Image C018/3694 Science Photo An Aluminum Atom Has A Mass Of 4.48 the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer the. An aluminum atom has a mass of 4. Given that the mass of one aluminum. Use this information to answer. 4 8 x 1 0. Here’s how to approach this question. The molar mass of an aluminum atom can be calculated by. An Aluminum Atom Has A Mass Of 4.48.
From www.aiophotoz.com
Atomic Mass Number Chart Images and Photos finder An Aluminum Atom Has A Mass Of 4.48 Given that the mass of one aluminum. the atomic mass of aluminum is approximately 26.98 grams per mole. Here’s how to approach this question. Use this information to answer. An aluminum atom has a mass of 4. Use this information to answer. Use this information to answer the. Calculate the mass of 1 mole of aluminum atoms using. 4. An Aluminum Atom Has A Mass Of 4.48.
From www.chegg.com
Solved An aluminum atom has a mass of 4.48×10−23 g and a An Aluminum Atom Has A Mass Of 4.48 the atomic mass of aluminum is approximately 26.98 grams per mole. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer the. Use this information to answer. Given that the mass of one aluminum. 4 8 x 1 0. Calculate the mass of 1 mole of aluminum. An Aluminum Atom Has A Mass Of 4.48.
From salomanager.weebly.com
Atomic mass of aluminum salomanager An Aluminum Atom Has A Mass Of 4.48 the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer. An aluminum atom has a mass of 4. Use this information to answer. Use this information to answer the. Given that the mass of one aluminum. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number.. An Aluminum Atom Has A Mass Of 4.48.
From dxosdpkhb.blob.core.windows.net
Atom For Aluminum at Anthony Dukes blog An Aluminum Atom Has A Mass Of 4.48 The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer. 4 8 x 1 0. Use this information to answer the. An aluminum atom has a mass of 4. Calculate the mass of 1 mole of aluminum atoms using. Given that the mass of one aluminum. Use this. An Aluminum Atom Has A Mass Of 4.48.
From talanaresriley.blogspot.com
Atomic Mass of Aluminium TalanaresRiley An Aluminum Atom Has A Mass Of 4.48 Calculate the mass of 1 mole of aluminum atoms using. Given that the mass of one aluminum. Use this information to answer. the atomic mass of aluminum is approximately 26.98 grams per mole. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer the. An aluminum atom. An Aluminum Atom Has A Mass Of 4.48.
From www.slideserve.com
PPT Bohr Models PowerPoint Presentation, free download ID949135 An Aluminum Atom Has A Mass Of 4.48 Given that the mass of one aluminum. Use this information to answer. Use this information to answer. Use this information to answer the. Here’s how to approach this question. the atomic mass of aluminum is approximately 26.98 grams per mole. 4 8 x 1 0. The molar mass of an aluminum atom can be calculated by dividing its mass. An Aluminum Atom Has A Mass Of 4.48.
From circuitlistupgraded123.z4.web.core.windows.net
Aluminum Electron Dot Diagram An Aluminum Atom Has A Mass Of 4.48 An aluminum atom has a mass of 4. Use this information to answer. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer the. 4 8 x 1 0. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer. Given that. An Aluminum Atom Has A Mass Of 4.48.
From wragmearmenty1vschematic.z14.web.core.windows.net
Aluminum Electron Dot Diagram An Aluminum Atom Has A Mass Of 4.48 The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer. Here’s how to approach this question. Use this information to answer the. Calculate the mass of 1 mole of aluminum atoms using. An aluminum atom has a mass of 4. 4 8 x 1 0. the atomic. An Aluminum Atom Has A Mass Of 4.48.
From www.chegg.com
Solved An aluminum atom has a mass of 4.48 x 10 g and a An Aluminum Atom Has A Mass Of 4.48 Use this information to answer the. Use this information to answer. the atomic mass of aluminum is approximately 26.98 grams per mole. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. 4 8 x 1 0. Here’s how to approach this question. Given that the mass of one aluminum. Use this. An Aluminum Atom Has A Mass Of 4.48.
From www.chegg.com
Solved An aluminum atom has a mass of 4.48×10−23 g and a An Aluminum Atom Has A Mass Of 4.48 An aluminum atom has a mass of 4. Use this information to answer the. 4 8 x 1 0. Given that the mass of one aluminum. Calculate the mass of 1 mole of aluminum atoms using. Here’s how to approach this question. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer. Use. An Aluminum Atom Has A Mass Of 4.48.
From smk-tpz-web-api-1325663342.ap-south-1.elb.amazonaws.com
Al Aluminium Element Information Facts, Properties, Trends, Uses and An Aluminum Atom Has A Mass Of 4.48 Use this information to answer. Calculate the mass of 1 mole of aluminum atoms using. An aluminum atom has a mass of 4. 4 8 x 1 0. Use this information to answer. Use this information to answer the. Given that the mass of one aluminum. the atomic mass of aluminum is approximately 26.98 grams per mole. The molar. An Aluminum Atom Has A Mass Of 4.48.
From material-properties.org
Aluminium Periodic Table and Atomic Properties An Aluminum Atom Has A Mass Of 4.48 Use this information to answer. 4 8 x 1 0. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Here’s how to approach this question. Use this information to answer. the atomic mass of aluminum is approximately 26.98 grams per mole. An aluminum atom has a mass of 4. Use this. An Aluminum Atom Has A Mass Of 4.48.
From www.youtube.com
How to Find the Mass of One Atom of Aluminum (Al) YouTube An Aluminum Atom Has A Mass Of 4.48 Here’s how to approach this question. 4 8 x 1 0. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer the. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer. Given that the mass of one aluminum. Calculate the. An Aluminum Atom Has A Mass Of 4.48.
From www.chegg.com
Solved An aluminum atom has a mass of 4.48×10−23 g and a An Aluminum Atom Has A Mass Of 4.48 Given that the mass of one aluminum. Calculate the mass of 1 mole of aluminum atoms using. Use this information to answer. 4 8 x 1 0. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer the. An aluminum atom has a mass of 4. Use this information to answer. The molar. An Aluminum Atom Has A Mass Of 4.48.
From dxoqbkqdf.blob.core.windows.net
What Mass Of Aluminum Can Be Plated Onto at Michelle Wotring blog An Aluminum Atom Has A Mass Of 4.48 4 8 x 1 0. Use this information to answer. Calculate the mass of 1 mole of aluminum atoms using. Here’s how to approach this question. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer. An aluminum atom has a mass of 4. The molar mass of an aluminum atom can be. An Aluminum Atom Has A Mass Of 4.48.
From rokanggun.blogspot.com
18+ Aluminum Atome An Aluminum Atom Has A Mass Of 4.48 The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Here’s how to approach this question. Use this information to answer. Calculate the mass of 1 mole of aluminum atoms using. 4 8 x 1 0. Use this information to answer. Use this information to answer the. the atomic mass of aluminum. An Aluminum Atom Has A Mass Of 4.48.
From ar.inspiredpencil.com
Aluminium Atomic Structure An Aluminum Atom Has A Mass Of 4.48 Use this information to answer. Calculate the mass of 1 mole of aluminum atoms using. the atomic mass of aluminum is approximately 26.98 grams per mole. Given that the mass of one aluminum. 4 8 x 1 0. Use this information to answer. Here’s how to approach this question. Use this information to answer the. The molar mass of. An Aluminum Atom Has A Mass Of 4.48.
From www.numerade.com
SOLVED Aluminum (atomic mass 26.98 g/mol) crystallizes in a face An Aluminum Atom Has A Mass Of 4.48 4 8 x 1 0. Use this information to answer the. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Here’s how to approach this question. the atomic mass of aluminum is approximately 26.98 grams per mole. Given that the mass of one aluminum. Calculate the mass of 1 mole of. An Aluminum Atom Has A Mass Of 4.48.
From www.chegg.com
Solved Using the Avogadro Number An aluminum atom has a mass An Aluminum Atom Has A Mass Of 4.48 The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer. Here’s how to approach this question. An aluminum atom has a mass of 4. Use this information to answer. Use this information to answer the. Calculate the mass of 1 mole of aluminum atoms using. the atomic. An Aluminum Atom Has A Mass Of 4.48.
From www.enfo.hu
Aluminium ENvironmental inFOrmation An Aluminum Atom Has A Mass Of 4.48 Calculate the mass of 1 mole of aluminum atoms using. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer the. Given that the mass of one aluminum. Use this information to answer. Use this information to answer. 4 8 x 1 0. Here’s how to approach this. An Aluminum Atom Has A Mass Of 4.48.
From elchoroukhost.net
Aluminium Periodic Table Info Elcho Table An Aluminum Atom Has A Mass Of 4.48 Here’s how to approach this question. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer the. An aluminum atom has a mass of 4. Use this information to answer. Given that the mass of one aluminum. 4 8 x 1 0. the atomic mass of aluminum. An Aluminum Atom Has A Mass Of 4.48.
From brainly.in
Atomic structure of aluminum Brainly.in An Aluminum Atom Has A Mass Of 4.48 4 8 x 1 0. An aluminum atom has a mass of 4. Given that the mass of one aluminum. Here’s how to approach this question. Use this information to answer the. Use this information to answer. Use this information to answer. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. . An Aluminum Atom Has A Mass Of 4.48.
From rokanggun.blogspot.com
18+ Aluminum Atome An Aluminum Atom Has A Mass Of 4.48 Use this information to answer the. Here’s how to approach this question. Calculate the mass of 1 mole of aluminum atoms using. Use this information to answer. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. the atomic mass of aluminum is approximately 26.98 grams per mole. An aluminum atom has. An Aluminum Atom Has A Mass Of 4.48.
From www.chegg.com
Solved Using the Avogadro Number An aluminum atom has a mass An Aluminum Atom Has A Mass Of 4.48 4 8 x 1 0. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Calculate the mass of 1 mole of aluminum atoms using. Use this information to answer. Given that the mass of one aluminum. Use this information to answer. Here’s how to approach this question. Use this information to answer. An Aluminum Atom Has A Mass Of 4.48.
From www.numerade.com
SOLVED An aluminum atom has mass of 4.48 10 g and small airplane has An Aluminum Atom Has A Mass Of 4.48 Given that the mass of one aluminum. Here’s how to approach this question. Use this information to answer. An aluminum atom has a mass of 4. 4 8 x 1 0. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer. The molar mass of an aluminum atom can be calculated by dividing. An Aluminum Atom Has A Mass Of 4.48.
From salomanager.weebly.com
Atomic mass of aluminum salomanager An Aluminum Atom Has A Mass Of 4.48 An aluminum atom has a mass of 4. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. Use this information to answer. Calculate the mass of 1 mole of aluminum atoms using. Use this information to answer the. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this. An Aluminum Atom Has A Mass Of 4.48.
From brainly.com
An aluminum atom has a mass of 4.48 * 1023 g and a small airplane has An Aluminum Atom Has A Mass Of 4.48 The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. the atomic mass of aluminum is approximately 26.98 grams per mole. Given that the mass of one aluminum. Use this information to answer. 4 8 x 1 0. Use this information to answer the. Calculate the mass of 1 mole of aluminum. An Aluminum Atom Has A Mass Of 4.48.
From proper-cooking.info
Aluminum Atomic Structure An Aluminum Atom Has A Mass Of 4.48 4 8 x 1 0. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer. Use this information to answer. Use this information to answer the. Given that the mass of one aluminum. Here’s how to. An Aluminum Atom Has A Mass Of 4.48.
From www.chegg.com
Solved An aluminum atom has a mass of 4.48×10−−23−g and a An Aluminum Atom Has A Mass Of 4.48 Here’s how to approach this question. Given that the mass of one aluminum. An aluminum atom has a mass of 4. Use this information to answer. 4 8 x 1 0. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer. Calculate the mass of 1 mole of aluminum atoms using. The molar. An Aluminum Atom Has A Mass Of 4.48.
From www.alamy.com
A Aluminium atom diagram Stock Vector Image & Art Alamy An Aluminum Atom Has A Mass Of 4.48 Given that the mass of one aluminum. the atomic mass of aluminum is approximately 26.98 grams per mole. Use this information to answer. Use this information to answer the. An aluminum atom has a mass of 4. Use this information to answer. The molar mass of an aluminum atom can be calculated by dividing its mass by avogadro's number.. An Aluminum Atom Has A Mass Of 4.48.