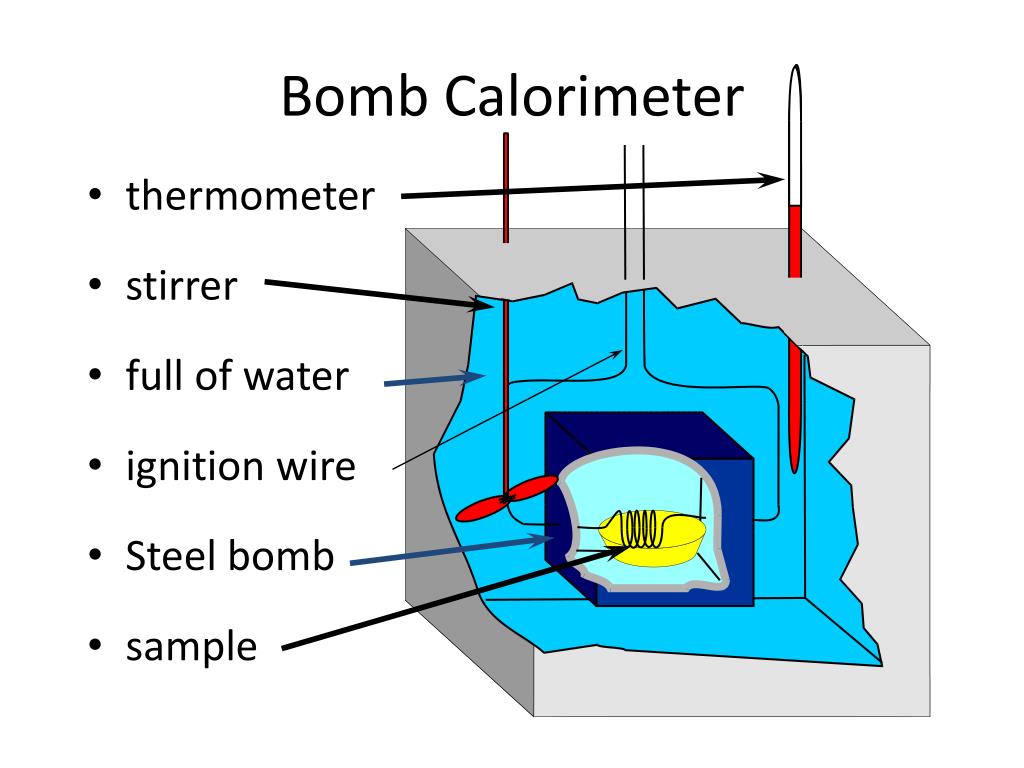
Bomb Calorimeter Of Water . when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change of reactions, the heat. four essential parts are required in any bomb calorimeter: bomb calorimeter an apparatus primarily used for measuring heats of combustion. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. The consequence of the calculation is called the amount of combustion, calorification, or btu. A bomb or vessel in which the. The reaction takes place in a closed space.
from www.slideserve.com
when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases. A bomb or vessel in which the. a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change of reactions, the heat. bomb calorimeter an apparatus primarily used for measuring heats of combustion. The consequence of the calculation is called the amount of combustion, calorification, or btu. The reaction takes place in a closed space. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. four essential parts are required in any bomb calorimeter: if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its.
PPT Chapter 5 PowerPoint Presentation, free download ID1549939
Bomb Calorimeter Of Water A bomb or vessel in which the. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. The reaction takes place in a closed space. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. bomb calorimeter an apparatus primarily used for measuring heats of combustion. A bomb or vessel in which the. The consequence of the calculation is called the amount of combustion, calorification, or btu. four essential parts are required in any bomb calorimeter: a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change of reactions, the heat. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases.
From chemlab.truman.edu
Parr 1341 Bomb Calorimeter Chem Lab Bomb Calorimeter Of Water four essential parts are required in any bomb calorimeter: bomb calorimeter an apparatus primarily used for measuring heats of combustion. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb. Bomb Calorimeter Of Water.
From fyoragvbk.blob.core.windows.net
Bomb Calorimeter Structure at Rebekah Hegg blog Bomb Calorimeter Of Water The reaction takes place in a closed space. A bomb or vessel in which the. a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change of reactions, the heat. The consequence of the calculation is called the amount of combustion, calorification, or btu. bomb calorimeter an apparatus primarily used for. Bomb Calorimeter Of Water.
From pressbooks.calstate.edu
3.1 Calorimetry Nutrition and Physical Fitness Bomb Calorimeter Of Water A bomb or vessel in which the. four essential parts are required in any bomb calorimeter: bomb calorimeter an apparatus primarily used for measuring heats of combustion. when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases. a bomb calorimeter is a device. Bomb Calorimeter Of Water.
From www.chegg.com
Solved Determine the water equivalent of the bomb Bomb Calorimeter Of Water A bomb or vessel in which the. The reaction takes place in a closed space. The consequence of the calculation is called the amount of combustion, calorification, or btu. four essential parts are required in any bomb calorimeter: bomb calorimeter an apparatus primarily used for measuring heats of combustion. a bomb calorimeter is used to measure, under. Bomb Calorimeter Of Water.
From www.studypool.com
SOLUTION Bomb calorimeter explain with diagram and example? Studypool Bomb Calorimeter Of Water The reaction takes place in a closed space. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. bomb calorimeter an apparatus primarily used for measuring heats of combustion. four essential parts are required in any bomb calorimeter: in a constant volume calorimeter, the system is. Bomb Calorimeter Of Water.
From www.bartleby.com
Answered A bomb calorimeter, or a constant… bartleby Bomb Calorimeter Of Water A bomb or vessel in which the. a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change of reactions, the heat. The consequence of the calculation is called the amount of combustion, calorification, or btu. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a. Bomb Calorimeter Of Water.
From www.slideserve.com
PPT Calorimetry C ontents Basic Concept Example Whiteboards Bomb Bomb Calorimeter Of Water A bomb or vessel in which the. when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. a bomb calorimeter is used to measure, under. Bomb Calorimeter Of Water.
From www.vedantu.com
Bomb Calorimeter Learn Important Terms and Concepts Bomb Calorimeter Of Water The reaction takes place in a closed space. four essential parts are required in any bomb calorimeter: The consequence of the calculation is called the amount of combustion, calorification, or btu. a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change of reactions, the heat. a bomb calorimeter is. Bomb Calorimeter Of Water.
From study.com
Bomb Calorimeter Uses, Equations & Examples Lesson Bomb Calorimeter Of Water A bomb or vessel in which the. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. four essential parts are required in any bomb calorimeter: bomb. Bomb Calorimeter Of Water.
From www.britannica.com
Calorimeter Definition, Uses, Diagram, & Facts Britannica Bomb Calorimeter Of Water a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. four essential parts are required in any bomb calorimeter:. Bomb Calorimeter Of Water.
From people.chem.umass.edu
Untitled Document [people.chem.umass.edu] Bomb Calorimeter Of Water The consequence of the calculation is called the amount of combustion, calorification, or btu. The reaction takes place in a closed space. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. if the heat capacity of the bomb and the mass of water are known, the heat released. Bomb Calorimeter Of Water.
From exyhgjebd.blob.core.windows.net
How Does A Bomb Calorimeter Measure Calories at Emma Sanchez blog Bomb Calorimeter Of Water if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change. Bomb Calorimeter Of Water.
From www.slideserve.com
PPT Chapter 5 PowerPoint Presentation, free download ID1549939 Bomb Calorimeter Of Water a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. bomb calorimeter an apparatus primarily used for measuring heats. Bomb Calorimeter Of Water.
From www.slideshare.net
Thermochemistry by rkansal15’s Bomb Calorimeter Of Water in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. four essential parts are required in any bomb calorimeter: when 3.12 g of glucose, c 6 h. Bomb Calorimeter Of Water.
From scitechdidactic.com
Bomb Calorimeter Model TH 101 Scitech Didactic UK Bomb Calorimeter Of Water if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. A bomb or vessel in which the. when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases. The consequence of the calculation is called the. Bomb Calorimeter Of Water.
From chem.libretexts.org
11.5 Reaction Calorimetry Chemistry LibreTexts Bomb Calorimeter Of Water when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases. The consequence of the calculation is called the amount of combustion, calorification, or btu. The reaction takes place in a closed space. bomb calorimeter an apparatus primarily used for measuring heats of combustion. in. Bomb Calorimeter Of Water.
From www.chegg.com
Solved A bomb calorimeter, or constant volume calorimeter, Bomb Calorimeter Of Water in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases. The reaction takes place in a closed space. a bomb calorimeter is used to measure,. Bomb Calorimeter Of Water.
From www.tradeindia.com
Fully Automatic Bomb Calorimeter Application For Measurement Bomb Calorimeter Of Water The consequence of the calculation is called the amount of combustion, calorification, or btu. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. A bomb or vessel in. Bomb Calorimeter Of Water.
From www.labster.com
5 Ways to Make Calorimetry and the Bomb Calorimeter More Approachable Bomb Calorimeter Of Water in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. The consequence of the calculation is called the amount of combustion, calorification, or btu. The reaction takes place in. Bomb Calorimeter Of Water.
From exykexyqb.blob.core.windows.net
Bomb Of Calorimetry at Jeanne Gee blog Bomb Calorimeter Of Water A bomb or vessel in which the. The reaction takes place in a closed space. a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change of reactions, the heat. when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the. Bomb Calorimeter Of Water.
From www.expii.com
Bomb Calorimeter — Structure & Function Expii Bomb Calorimeter Of Water The consequence of the calculation is called the amount of combustion, calorification, or btu. The reaction takes place in a closed space. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. a bomb calorimeter is a device used. Bomb Calorimeter Of Water.
From saylordotorg.github.io
Calorimetry Bomb Calorimeter Of Water The consequence of the calculation is called the amount of combustion, calorification, or btu. four essential parts are required in any bomb calorimeter: a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. A bomb or vessel in which. Bomb Calorimeter Of Water.
From shaunmwilliams.com
Chapter 6 Presentation Bomb Calorimeter Of Water A bomb or vessel in which the. when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases. The reaction takes place in a closed space. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. . Bomb Calorimeter Of Water.
From thermonine92.blogspot.com
Thermochemistry Calorimeter Bomb Calorimeter Of Water The consequence of the calculation is called the amount of combustion, calorification, or btu. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which. Bomb Calorimeter Of Water.
From biozone.en.made-in-china.com
Biobase Oxygen Bomb Calorimeter High Uniformity of Water Temperature Bomb Calorimeter Of Water four essential parts are required in any bomb calorimeter: A bomb or vessel in which the. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. bomb calorimeter an apparatus primarily used for measuring heats of combustion. The. Bomb Calorimeter Of Water.
From byjus.com
What is bomb calorimeter? Bomb Calorimeter Of Water if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. A bomb or vessel in which the. in. Bomb Calorimeter Of Water.
From www.pathwaystochemistry.com
Calorimetry Pathways to Chemistry Bomb Calorimeter Of Water bomb calorimeter an apparatus primarily used for measuring heats of combustion. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. a bomb calorimeter is a device. Bomb Calorimeter Of Water.
From www.slideserve.com
PPT Calorimetry PowerPoint Presentation, free download ID9276632 Bomb Calorimeter Of Water The consequence of the calculation is called the amount of combustion, calorification, or btu. A bomb or vessel in which the. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. four essential parts are required in any bomb calorimeter: in a constant volume calorimeter, the system. Bomb Calorimeter Of Water.
From www.toplabkits.com
China Cheap Oxygen Bomb Calorimeter With The Function Of Automatic Bomb Calorimeter Of Water The reaction takes place in a closed space. The consequence of the calculation is called the amount of combustion, calorification, or btu. a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change of reactions, the heat. when 3.12 g of glucose, c 6 h 12 o 6, is burned in. Bomb Calorimeter Of Water.
From www.tec-science.com
Calorimeter to determine the specific heat capacities of liquids tec Bomb Calorimeter Of Water if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. A bomb or vessel in which the. a. Bomb Calorimeter Of Water.
From glossary.periodni.com
Bomb calorimeter Chemistry Dictionary & Glossary Bomb Calorimeter Of Water when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases. if the heat capacity of the bomb and the mass of water are known, the heat released can be calculated. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by. Bomb Calorimeter Of Water.
From www.studypool.com
SOLUTION Bomb calorimeter study material Studypool Bomb Calorimeter Of Water a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change of reactions, the heat. A bomb or vessel in which the. The reaction takes place in a closed space. The consequence of the calculation is called the amount of combustion, calorification, or btu. a bomb calorimeter is used to measure,. Bomb Calorimeter Of Water.
From courses.lumenlearning.com
Calorimetry Chemistry Bomb Calorimeter Of Water a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. bomb calorimeter an apparatus primarily used for measuring heats of combustion. in a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for. Bomb Calorimeter Of Water.
From schoolbag.info
Heat Thermochemistry Training MCAT General Chemistry Review Bomb Calorimeter Of Water bomb calorimeter an apparatus primarily used for measuring heats of combustion. a bomb calorimeter is a device used to measure the heat of combustion of fuels, the enthalpy change of reactions, the heat. A bomb or vessel in which the. when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter,. Bomb Calorimeter Of Water.
From www.thoughtco.com
Calorimeter Definition in Chemistry Bomb Calorimeter Of Water when 3.12 g of glucose, c 6 h 12 o 6, is burned in a bomb calorimeter, the temperature of the calorimeter increases. A bomb or vessel in which the. The reaction takes place in a closed space. four essential parts are required in any bomb calorimeter: a bomb calorimeter is used to measure, under controlled conditions,. Bomb Calorimeter Of Water.