Understanding how to calculate units for 'k' is essential for precision in scientific, technical, and industrial calculations—whether converting scientific constants, engineering measurements, or financial metrics. This guide explains the core principles behind unit conversion and how to apply them effectively.

Understanding the Role of Unit 'k' in Measurements
In many contexts, 'k' represents a unit multiplier, often denoting thousands (e.g., 1 kΩ = 1000 ohms). To calculate units for 'k', identify the base unit and apply the scale factor consistently. For example, converting kN (kilonewtons) to newtons requires multiplying by 1000. Accurate interpretation of 'k' ensures correct dimensional analysis and avoids costly errors in calculations.

Step-by-Step Method for Calculating Units for k
Begin by isolating the value associated with 'k'. Multiply by the conversion factor—typically 1000 for SI-derived units—ensuring dimensional consistency. For instance, if force is given as 5 kN, calculate 5 × 1000 = 5000 N. Verify units remain coherent: multiplying kilograms by meters per second squared yields kilogram-meters per second squared (kg·m/s²), the correct unit for force. Always check dimensional formulas before finalizing results.
Common Applications and Practical Examples
Calculating units for 'k' is vital in engineering, physics, and data science. In electronics, converting kΩ to Ω enables proper circuit design. In climate modeling, scaling temperature changes from kelvins to degrees ensures accurate predictions. For financial modeling, adjusting k-values in growth projections maintains numerical integrity. Practice with real datasets to build confidence—convert 2.5 kWh to joules by multiplying by 3.6 million to maintain precision.
Mastering the calculation of units for 'k' enhances accuracy across disciplines. Use clear conversion factors, verify dimensional consistency, and apply these principles to real-world problems. Begin practicing today—precision starts with understanding the units.

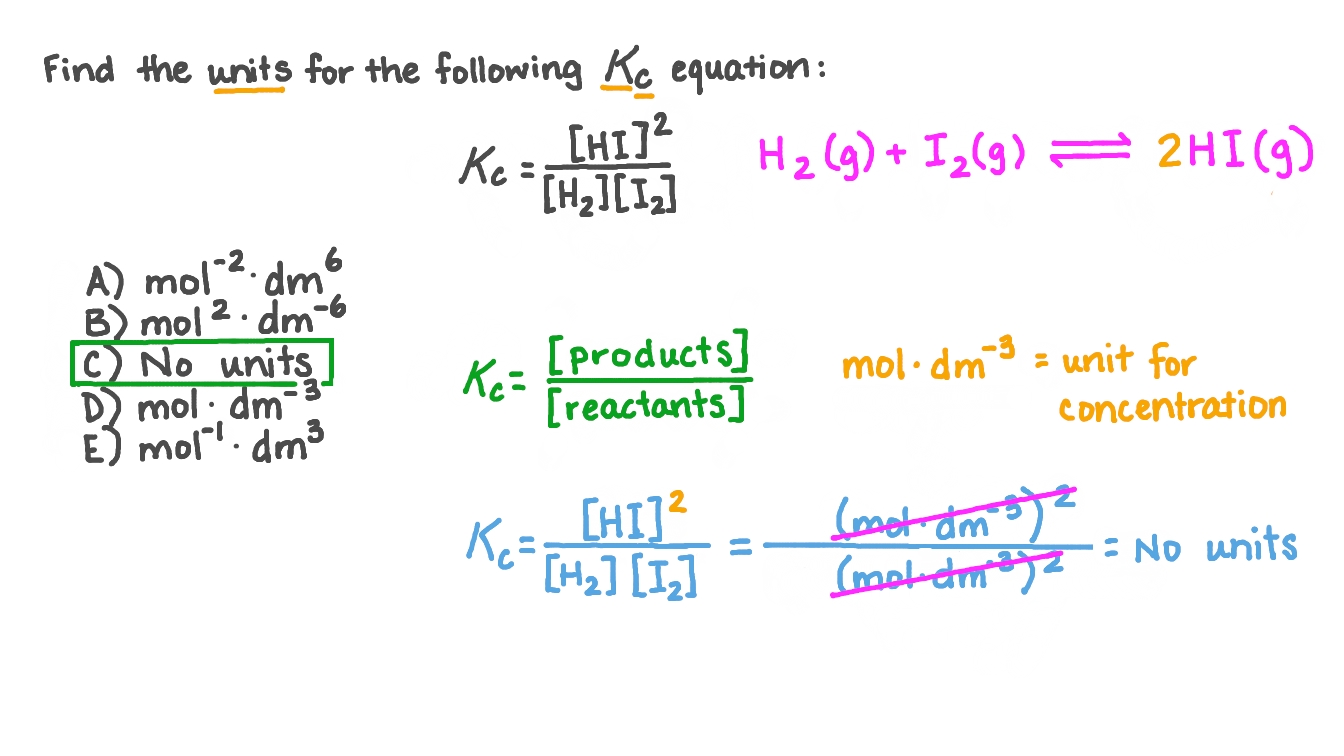
From this equation, a general formula for the units of k is obtained which is: k units = M1-n t-1 where n is the reaction order For example, let's say we want to determine the units of the rate constant for third-order reactions. n = 3, and therefore, k units = M1-3 t-1 = M-2 t-1 If the time is seconds, then the units will be. The rate constant is different for every reaction.

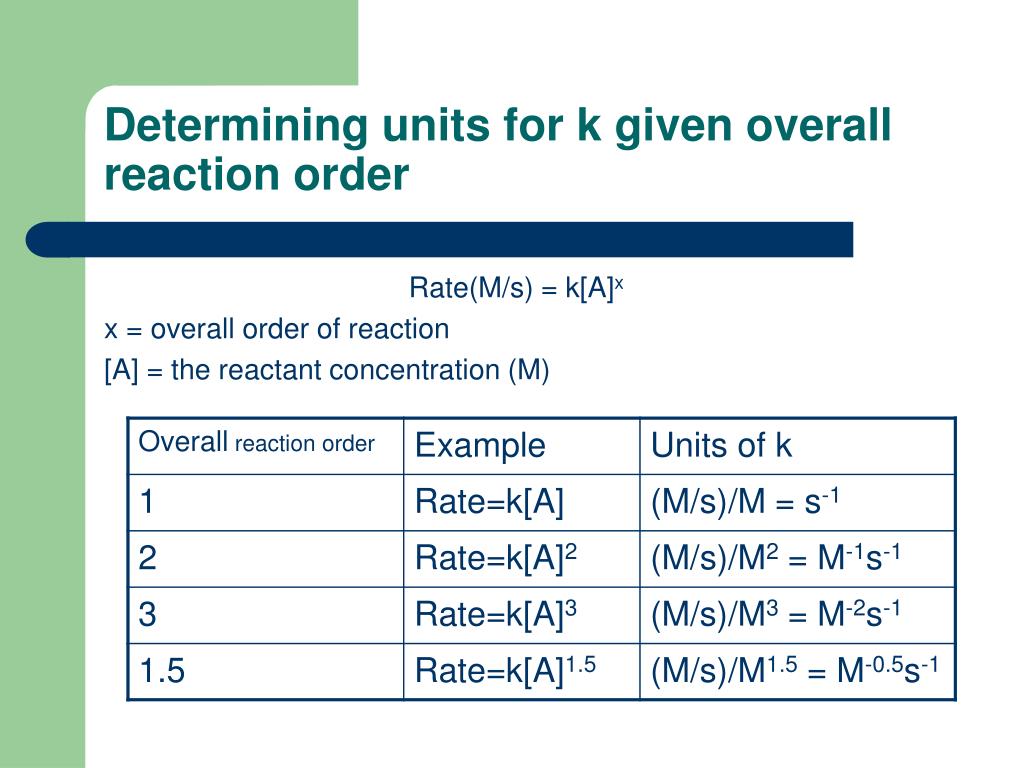
The larger the value of k, the faster the rate of reaction. The value of k can be determined by substituting in the values of rate and concentration of reactants into the rate equation, and then rearranging. The Units of the Rate Constant, k The units of k depend on the overall order of the reaction.
Zero Order Reaction A zero order reaction. This chemistry video tutorial explains how to determine the units of the rate constant K for a first order reaction, second order reaction, and a zero order reaction. It provides a formula and a.

The units for the rate of a reaction are mol/L/s. The units for k are whatever is needed so that substituting into the rate law expression affords the appropriate units for the rate. The units of the rate constant, k, depend on the overall reaction order.
The units of k for a zero-order reaction are M/s, the units of k for a first-order reaction are 1/s, and the units of k for a second-order reaction are 1/ (M s). Learn about rate constant calculations for your A. Choose the Time Unit: Select seconds, minutes, or hours as the unit of time.

Click "Calculate": The calculator will instantly display: Rate constant (k) Units for k General rate law Integrated rate law used for the calculation Click "Reset": This clears all fields and resets the calculator. Rate constant calculator to compute k from rate laws or integrated kinetics (0th/1st/2nd). Shows steps, mini chart, log-scale gauge, and unit handling.
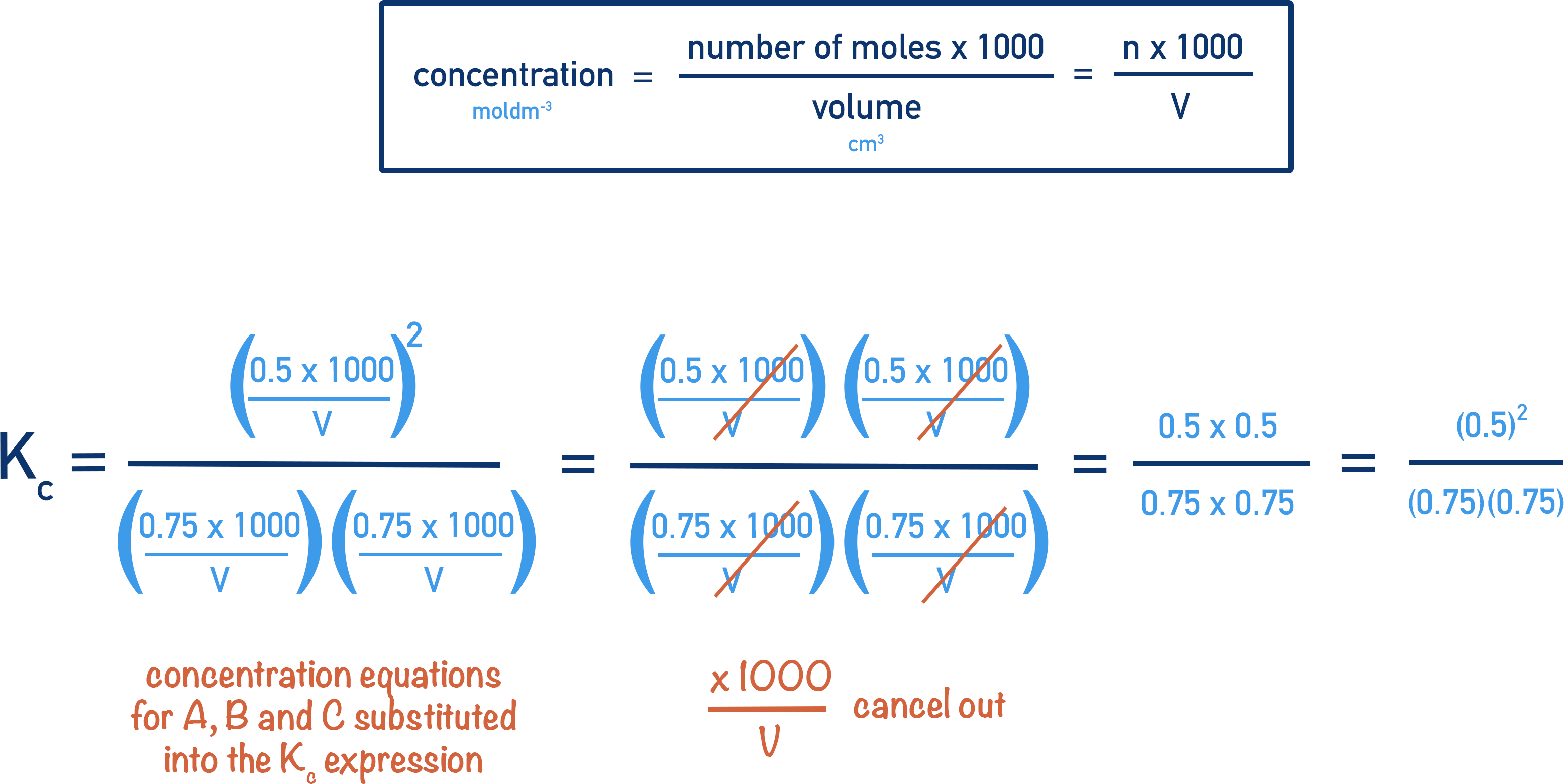
In this article, we will learn about rate constant, rate units, rate law, and integrated rate law equations of a different order. Calculating the Rate Constant To calculate k, use the rate equation: rate = k [A] [B] for a second-order reaction, where [A] and [B] are the molar concentrations of the reactants. The value of k can be isolated by rearranging this equation to k = \text {rate} / [A] [B].
Units of the Rate Constant The units of k vary with the reaction order.