This gallery shows what chemical elements look like before they form compounds with other elements. The elements are shown in the order they appear on the periodic table, starting with hydrogen. Some rare elements don't have pictures because they're highly radioactive and vanish quickly after creation.
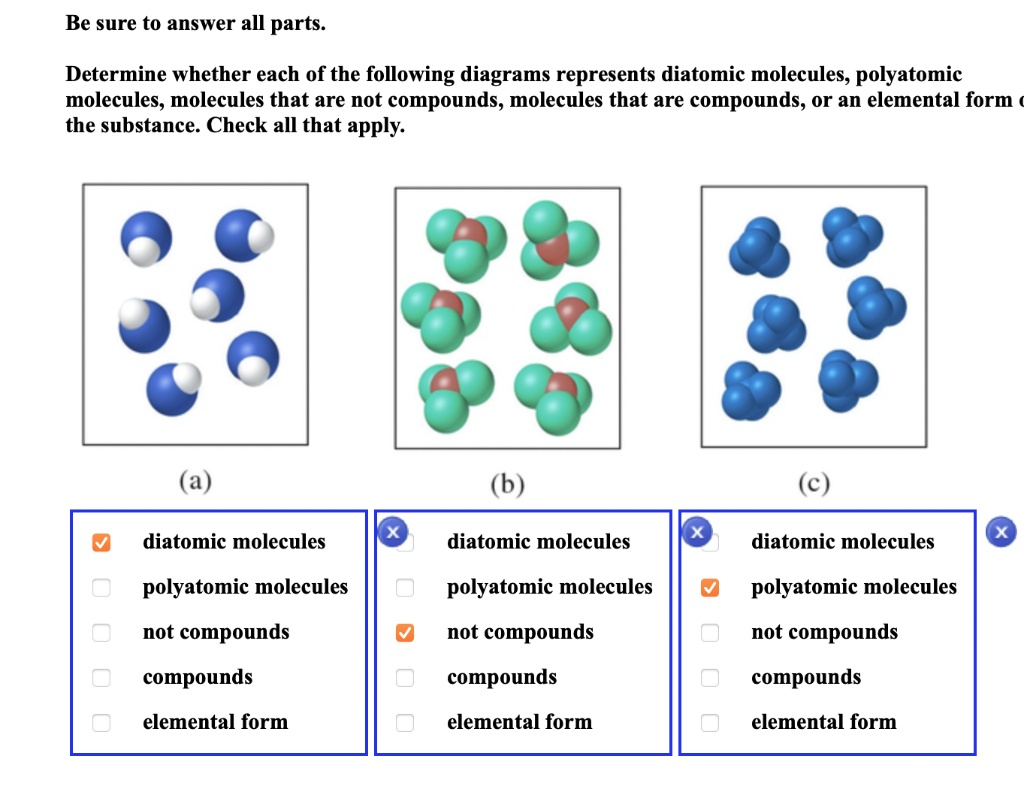
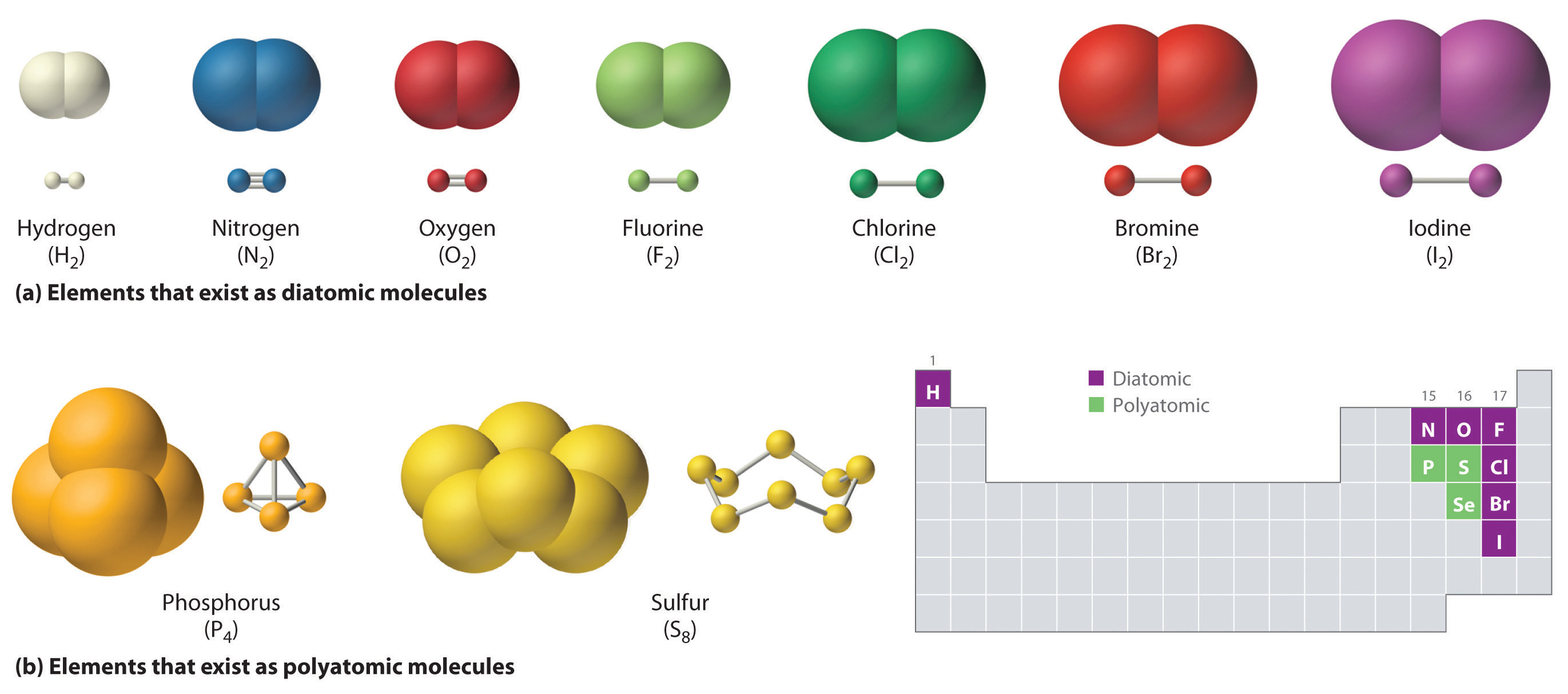
Master Periodic Table: Elemental Forms with free video lessons, step-by-step explanations, practice problems, examples, and FAQs. Learn from expert tutors and get exam-ready! Elements Any substance that contains only one kind of an atom is known as an element. Because atoms cannot be created or destroyed in a chemical reaction, elements such as phosphorus (P 4) or sulfur (S 8) cannot be broken down into simpler substances by these reactions.

A DIAGRAM OF AN ELEMENTAL FORM OF A SUBSTANCE - Brainly.in
Example: Water decomposes into a mixture of hydrogen and oxygen when an electric current is passed through the liquid. Elements An element is the simplest form of matter that has a unique set of properties. It is a pure substance that cannot be broken down into different types of substances.

As of 2010, there are 118 known elements. Ninety eight of the elements are naturally occurring on earth, while the other 20 elements have been made synthetically in a lab. Learning Objectives Define a chemical element and give examples of the abundance of different elements.

PPT - Ch 13. Group 13 PowerPoint Presentation, free download - ID:5491992
Represent a chemical element with a chemical symbol. State the periodic law and explain the organization of elements in the periodic table. Predict the general properties of elements based on their location within the periodic table.

Identify metals, nonmetals, and metalloids by their. Interactive periodic table showing names, electrons, and oxidation states. Visualize trends, 3D orbitals, isotopes, and mix compounds.

3.1: Types of Chemical Compounds and their Formulas - Chemistry LibreTexts
Fully descriptive writeups. The elements of the table are ordered by their atomic number. This number increases by 1 as you progress from element to element across the table.
We will discuss the significance of the atomic number in greater detail in Chapter 3 of the Chemistry Tutorial. Each cell of the periodic table also lists the atomic mass of each element. Element is the most basic form of matter (or a chemical substance) that exists under ordinary conditions.
Sodium, carbon, copper, silver, hydrogen, oxygen, nitrogen, etc., are some familiar examples of elements. The term elemental is used to describe the most stable physical state that an element exists in under normal conditions-normal temperatures and pressures. For example, the most stable form for chlorine atoms is as discrete diatomic structures, i.e.
two chlorine atoms bonded together comprise one unit or molecule of elemental chlorine. Chemical elements are the basic building blocks of matter. Elements are referred to by their names and their symbols; this makes it easier to write chemical structures and equations.
Examples of elements include hydrogen, helium, and lithium.