
September 5, 2024
Centrally Acting Medications For Weight Problems: Past, Present, Andfuture Pmc
Long-lasting Effectiveness And Safety Look at more info Of Anti-obesity Treatment: Where Do We Stand? Present Weight Problems Reports There is sufficient proof supporting that pharmacotherapy in mix with behavior-based treatments can cause considerable weight loss and enhanced cardiometabolism. Recalling with the background of obesity treatment, we note that thefirst reduced carb diet plan was the Banting Diet plan, released in 1863. Diet still plays a vital role inweight loss, but longterm pharmacotherapies with restricted negative effects are criticalfor keeping weight loss. The first jejunoileal bypass for excessive weight was reportedin the 1950's [128], and the operationdid not come to be prominent until the 1970's. More advanced treatments are usednow and surgery still has a significant place in the treatment of excessive weight, givingthe largest weight-loss, ideal maintenance of weight-loss, and reversal of insulinresistance. This section on future anti-obesity drugs focuses on tesofensine, since itis the only CNS acting anti-obesity drug that has actually reached an innovative phase ofdevelopment. Two of the four trials will be carried out for the weight problems studies each for a period of one year. The trials will certainly also consist of a two-year research to observe the safety and security and efficiency of the drug on the cardio system. Effectiveness researches deal with the concern of just how much additional weight decrease is a good idea in a finite duration, and the period essential for documenting it with self-confidence. Given the effectiveness that is being attained and the persistent nature of excessive weight, it is feasible that keeping the price in weight loss for topics of continued excess weight is the key purpose. Reducing the researches with the purpose of increasing the relative rate of weight reduction might not show a good idea for the person and could cause adverse effects that get rid of methods that otherwise would certainly verify sensible, if applied much less boldy. This is a factor of certain importance in the analysis of glucagon-based tri-agonists that intend to outperform GLP1-- GIPR co-agonists, as glucagon is likely an agonist of minimized healing index about both incretins.
Semaglutide
Both medications enhanced glycemic control, generated comparable fat burning, and minimized high blood pressure (55 ). One of the most regular adverse effects were transient moderate nausea or vomiting and small hypoglycemia, which were much less usual with liraglutide than with exenatide (56 ). Antibodies established with a lower frequency in liraglutide-treated subjects than in those treated by exenatide, likely because of its higher structural resemblance with human GLP-1 (97 vs. 52%). Nonetheless, it is urging that the development of antibodies does not impact the medicine efficacy.- PYY3-- 36 was located to have affordable bioavailability when given up a nasal spray formulation, yet in a 12-week, randomised, double-blind, placebo-controlled, scientific test in healthy overweight subjects, nasal management of the reduced dose produced only nominally better weight-loss than placebo.
- An even more detailed characterization of clients must offer to enhance the near-term likelihood for success and give enlightened direction for advancing the future generation of AOMs.
- At the time of diagnosis up to 90% of patients with craniopharyngioma are reported to have at the very least one pituitary hormonal agent shortage (39, 40, 50).
- The triple device of activity, nevertheless, might present major side-effect issues in large-scale trials.
What course of drug is tesofensine?
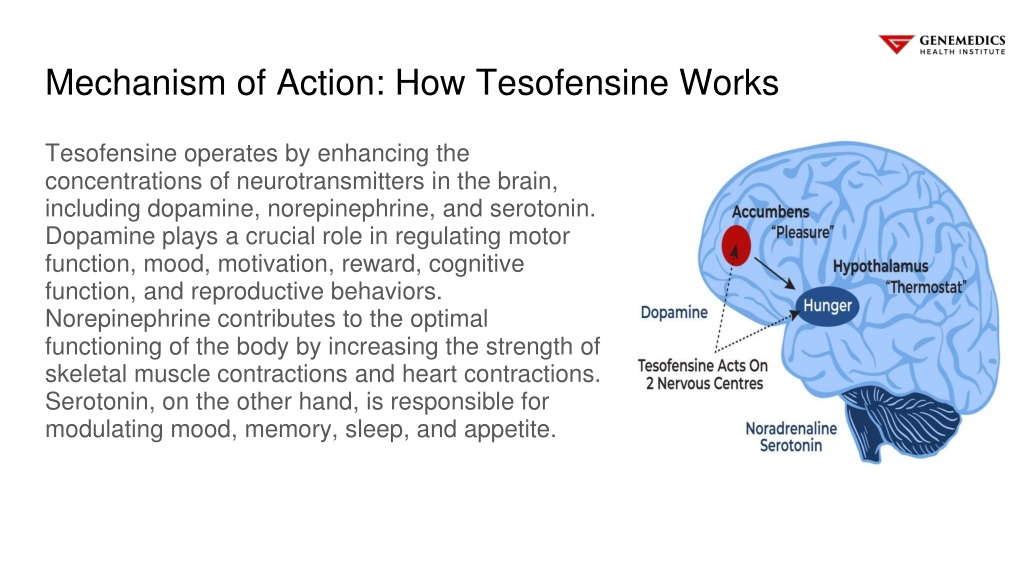
Tesofensine is a Serotonin-norepinephrine-dopamine-reuptake-inhibitor (SNDRI). SNDRIs are a class of psychedelic antidepressants. They act on neurotransmitters in the brain, namely, serotonin, norepinephrine and dopamine.
2 Semaglutide
In a stage II clinical trial, overweight clients got 0.25, 0.5, or 1 mg of tesofensine or sugar pill over 24 weeks after a 2 week run-in duration (Astrup et al., 2008). One of the most common damaging occasions were completely dry mouth, queasiness, irregularity, hard feceses, looseness of the bowels, and sleep problems. Boosts in heart rate and high blood pressure were additionally observed, which might restrict further boosts in dosing. The impacts of PSN S1 (Fig. 2) and PSN S2 on bodyweight and food intake were comparable in magnitude to those of sibutramine (Thomas et al., 2006). The weight-losses were moderated by a careful reduction in adiposity along with boosted insulin sensitivity, but plasma lipid profiles were not altered (Thomas et al., 2006). PSN S1 was ultimately taken into medical growth, however the program has actually currently been discontinued. An additional drug, Tesofensine, is a combined norepinephrine-serotonin-dopamine reuptake prevention currently under way for Phase 3 trials.Social Links