In the rapidly evolving world of digital avatars, Beyfortus stands out as a unique character whose age narrative blends design evolution with technological advancement—an age worth unpacking.
/what-is-beyfortus.jpg.aspx)
The Age of Beyfortus: A Timeline of Evolution
Beyfortus debuted with a distinct age classification that reflects both its conceptual roots and technical architecture. Developed in the mid-2020s, its initial design emphasized a youthful aesthetic paired with adaptive AI behaviors. Over time, updates have refined its age representation through dynamic character modeling, blending retro charm with modern responsiveness, making it a case study in responsive digital identity.

Decoding Age in Digital Personas
Age in virtual characters like Beyfortus isn't literal but symbolic—representing maturity in behavior, language, and interaction style. This age reflects user engagement patterns and AI learning milestones, offering insight into how digital beings evolve alongside audience expectations and platform capabilities.
Why Beyfortus’ Age Matters Today
Understanding Beyfortus’ age helps developers, designers, and fans anticipate how such characters will adapt to future trends. With rising demands for personalized and evolving digital experiences, age-based design informs smarter, more intuitive user journeys that foster long-term connection and relevance.
Beyfortus’ age encapsulates more than a timeline—it symbolizes the fusion of timeless appeal and cutting-edge innovation. As digital identities grow more sophisticated, recognizing these nuances empowers creators to build characters that resonate across generations. Dive deeper into the future of digital evolution today.
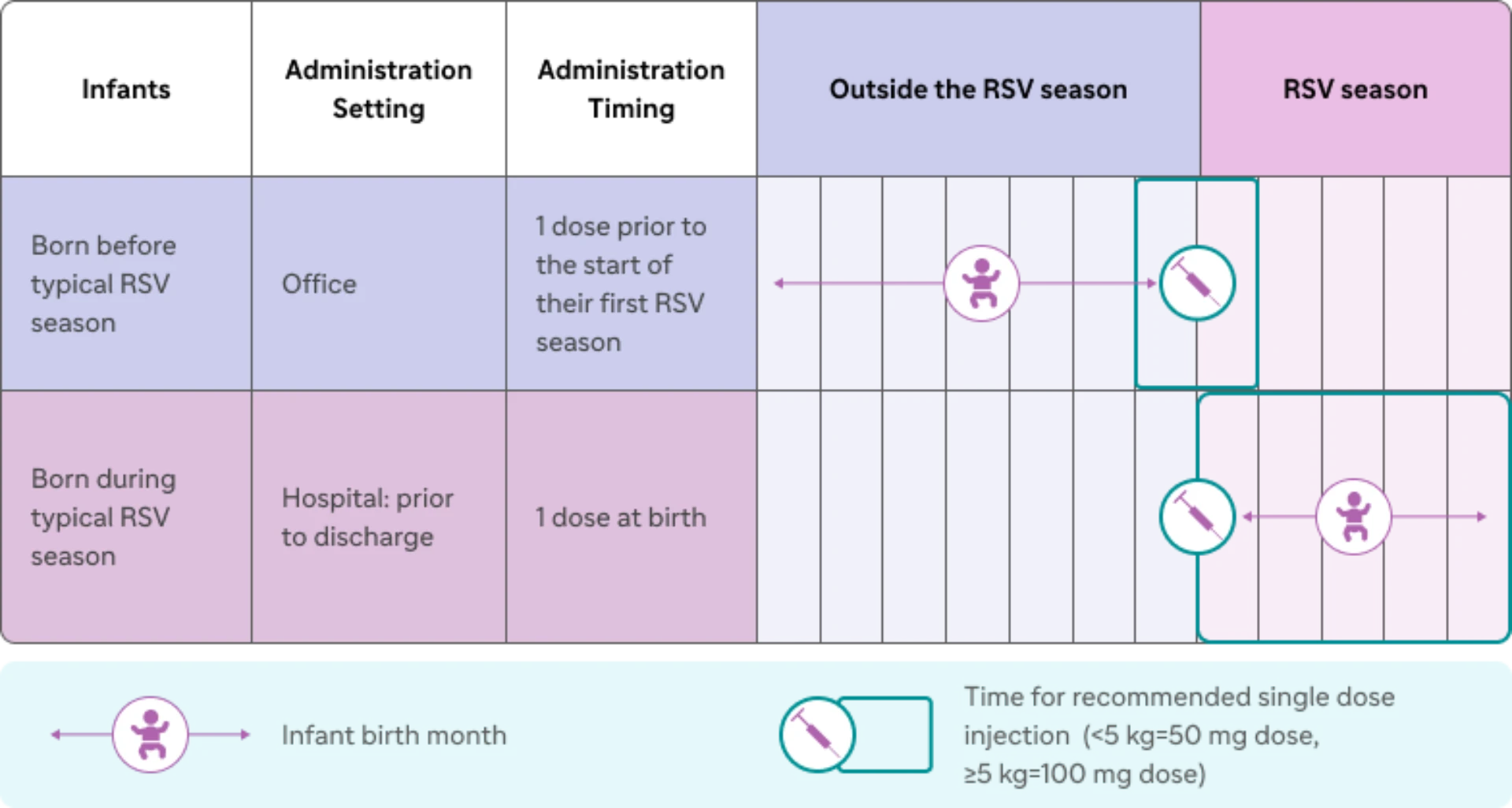
The child's age on the day the infant RSV antibody is administered should be used to determine if the child is eligible for immunization. Except in rare circumstances, an infant RSV antibody is not needed for most infants who are born 14 or more days after their mother received RSV vaccine. Beyfortus is indicated for the prevention of RSV lower respiratory tract disease in 1: Neonates and infants born during or entering their first RSV season.

Children up to 24 months of age who remain vulnerable to severe RSV disease through their second RSV season. Trial 05, a randomized, double-blind, active (palivizumab)-controlled, multicenter trial, supported the use of Beyfortus in in children up to 24 months of age who remain vulnerable to severe RSV. Beyfortus is a prescription medicine used to help prevent a serious lung disease caused by Respiratory Syncytial Virus (RSV) in: Newborns and babies under 1 year of age born during or entering their first RSV season.

Beyfortus is the first and only long-acting antibody indicated for the prevention of RSV lower respiratory tract disease in3: Neonates and infants born during or entering their first RSV season Children up to 24 months of age who remain vulnerable to severe RSV disease through their American Academy Pediatrics; ACIP. Beyfortus is used to prevent respiratory syncytial virus (RSV) lower respiratory tract disease in neonates and infants born during or entering their first RSV season. It may also be given to children up to 24 months of age who remain at risk of severe RSV disease through their second RSV season.

Beyfortus helps to protect healthy and at. Rx Only PATIENT INFORMATION BEYFORTUS® (Bay for tus) (nirsevimab-alip) injection, for intramuscular use What is BEYFORTUS? t is used to help prevent a serious lung disease caused by Respiratory Syncytial Virus (RSV newborns and babies under 1 year of age born during or entering their first RSV season. RSV Preventive Antibodies The RSV preventive antibody (generic name nirsevimab, trade name Beyfortus) is a shot that prevents severe RSV disease in infants and young children.

Antibodies are proteins that the body's immune system uses to fight off harmful germs. A total of 3,224 pediatric subjects received the recommended dose of BEYFORTUS in Phase 2 and Phase 3 clinical trials (Trials 03, 04, and 05) including 2,119 infants who were born at 35 weeks gestational age (GA) or older, and 1,105 infants who were born at less than 35 weeks GA. The Centers for Disease Control and Prevention's (CDC) Advisory Committee on Immunization Practices (ACIP) is recommending that Beyfortus ™ (nirsevimab-alip) be routinely used for the prevention of respiratory syncytial virus (RSV) lower respiratory tract disease for newborns and infants younger than 8 months of age born during or entering.
