Mastering Unit Calculation for k: A Step-by-Step Guide
In science and engineering, precise unit calculations are essential for accurate results. One common challenge involves determining the correct units for the constant k, which appears in formulas across physics, chemistry, and applied mathematics. Mastering how to calculate units for k ensures consistency, prevents costly errors, and enhances problem-solving efficiency.

www.slideserve.com
Understanding the Constant k and Its Unit Requirements
The constant k varies depending on the context—such as in Ohm’s Law, the ideal gas law, or radioactive decay—but always requires careful unit analysis. For instance, in Ohm’s Law (V = IR), k represents resistance, measured in ohms (Ω). When calculating k, identify its dimensional form and convert input units into the correct output units using conversion factors. This ensures dimensional compatibility, a cornerstone of scientific rigor.

www.youtube.com
Step-by-Step Process for Calculating Units of k
To calculate units for k, follow these steps: First, define k’s role in the equation and list its base dimensions (e.g., mass, length, time). Next, list the units of all known variables. Apply dimensional analysis by dividing k’s desired units by the units of the variables, canceling common terms and isolating k’s units. Use conversion factors when necessary—for example, converting grams to kilograms or seconds to hours—to express k in standard units. Always verify dimensional consistency to avoid misinterpretation.
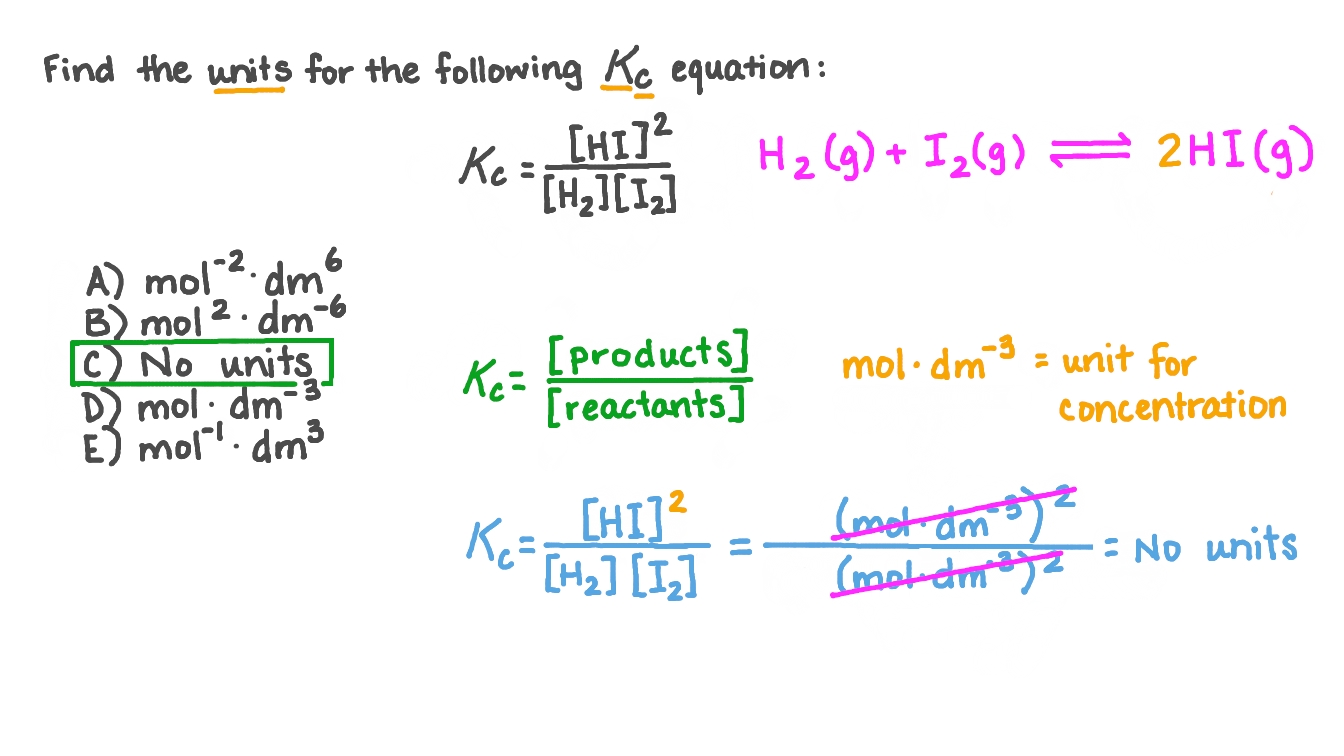
storage.googleapis.com
Common Mistakes and Best Practices in Unit Calculation for k
A frequent error is neglecting unit conversions, leading to incorrect results—such as treating k as dimensionless or mixing metric and imperial units. Always double-check dimensional balance, use conversion tables, and annotate units at each step. When working with complex equations, break the calculation into smaller parts to minimize mistakes. Practicing with real-world examples strengthens accuracy and confidence in unit handling for k.
www.slideserve.com
Calculating units for k is a fundamental skill that underpins precision in technical fields. By understanding dimensional relationships, applying systematic conversion methods, and avoiding common pitfalls, professionals can ensure reliable and reproducible results. Mastery of this concept not only enhances accuracy but also builds a strong foundation for advanced scientific and engineering applications.

www.slideserve.com
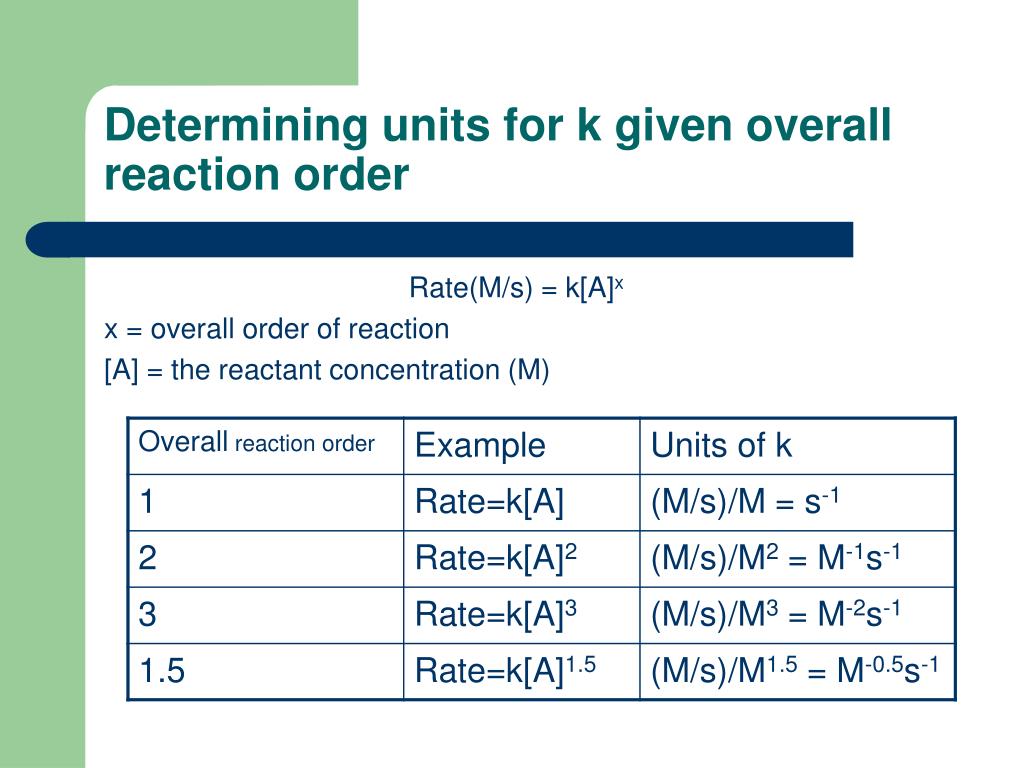
From this equation, a general formula for the units of k is obtained which is: k units = M1-n t-1 where n is the reaction order For example, let's say we want to determine the units of the rate constant for third-order reactions. n = 3, and therefore, k units = M1-3 t-1 = M-2 t-1 If the time is seconds, then the units will be. The units for the rate of a reaction are mol/L/s.

www.slideshare.net
The units for k are whatever is needed so that substituting into the rate law expression affords the appropriate units for the rate. The rate constant is different for every reaction. The larger the value of k, the faster the rate of reaction.
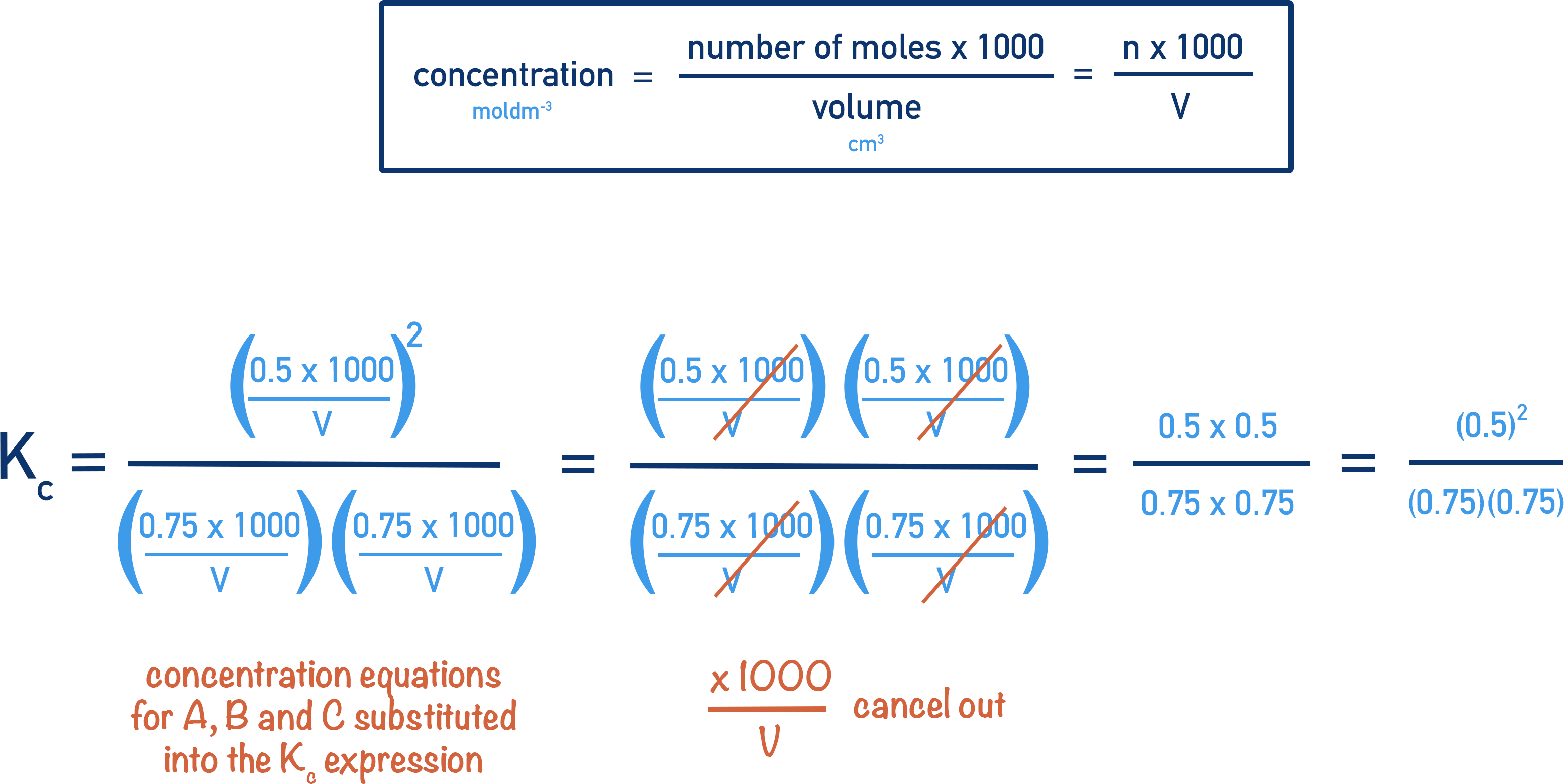
www.chemistrystudent.com
The value of k can be determined by substituting in the values of rate and concentration of reactants into the rate equation, and then rearranging. The Units of the Rate Constant, k The units of k depend on the overall order of the reaction. Zero Order Reaction A zero order reaction.

www.slideserve.com
This chemistry video tutorial explains how to determine the units of the rate constant K for a first order reaction, second order reaction, and a zero order reaction. It provides a formula and a. The units of the rate constant, k, depend on the overall reaction order.
The units of k for a zero-order reaction are M/s, the units of k for a first-order reaction are 1/s, and the units of k for a second-order reaction are 1/ (M s). Learn about rate constant calculations for your A. Choose the Time Unit: Select seconds, minutes, or hours as the unit of time.
Click "Calculate": The calculator will instantly display: Rate constant (k) Units for k General rate law Integrated rate law used for the calculation Click "Reset": This clears all fields and resets the calculator. Rate constant calculator to compute k from rate laws or integrated kinetics (0th/1st/2nd). Shows steps, mini chart, log-scale gauge, and unit handling.
Precisely calculating k is fundamental for process optimization, predictive modeling of reaction yields, and understanding reaction mechanisms. This article provides a comprehensive guide to calculating k, focusing on methodologies relevant to a technical audience. In this article, we will learn about rate constant, rate units, rate law, and integrated rate law equations of a different order.