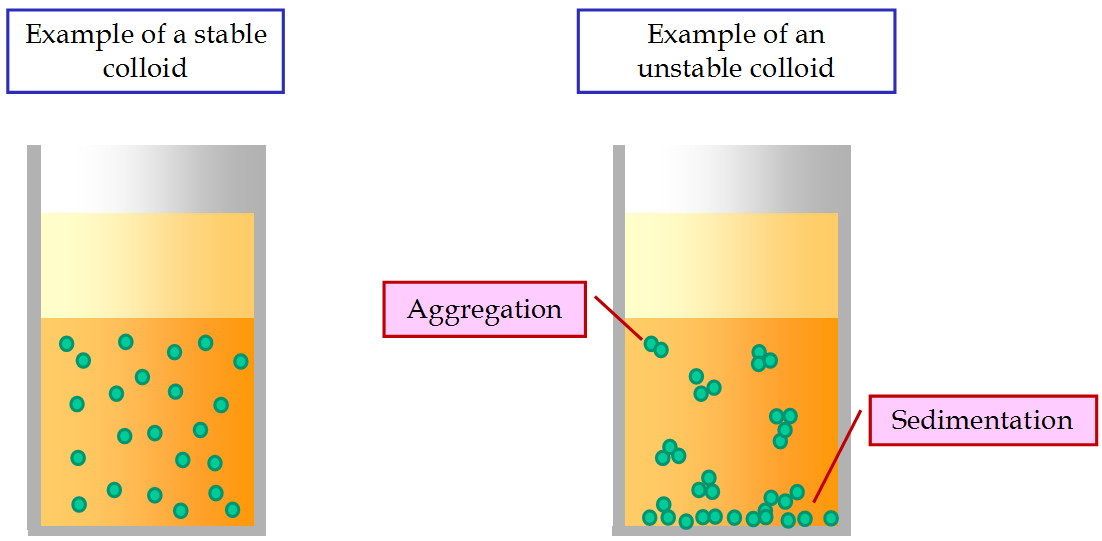
Opposite Of Suspension In Chemistry . Explore all the neet chemistry. — a suspension is a heterogeneous mixture in which particles disperse within a liquid or gas (fluid) but. — the suspension is a heterogeneous mixture in which the solute. The particles in suspensions are larger than those found in solutions. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. solutions are typically clear and transparent due to the small size of the solute particles. — the table below shows the main differences between suspension and emulsion: a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. This clarity allows light to pass through the solution without. Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out.
from byjus.com
— the suspension is a heterogeneous mixture in which the solute. This clarity allows light to pass through the solution without. a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. — a suspension is a heterogeneous mixture in which particles disperse within a liquid or gas (fluid) but. Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. The particles in suspensions are larger than those found in solutions. solutions are typically clear and transparent due to the small size of the solute particles. — the table below shows the main differences between suspension and emulsion: — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. Explore all the neet chemistry.
Suspensions (Chemistry) Definition, Properties, Examples with Videos
Opposite Of Suspension In Chemistry The particles in suspensions are larger than those found in solutions. Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. Explore all the neet chemistry. This clarity allows light to pass through the solution without. a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. — the table below shows the main differences between suspension and emulsion: — a suspension is a heterogeneous mixture in which particles disperse within a liquid or gas (fluid) but. — the suspension is a heterogeneous mixture in which the solute. The particles in suspensions are larger than those found in solutions. solutions are typically clear and transparent due to the small size of the solute particles. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase.
From www.slideserve.com
PPT Chapter 8 Solutions PowerPoint Presentation, free download ID Opposite Of Suspension In Chemistry — the table below shows the main differences between suspension and emulsion: — a suspension is a heterogeneous mixture in which particles disperse within a liquid or gas (fluid) but. This clarity allows light to pass through the solution without. — the suspension is a heterogeneous mixture in which the solute. — a suspension is a. Opposite Of Suspension In Chemistry.
From www.slideserve.com
PPT Properties of Matter PowerPoint Presentation, free download ID Opposite Of Suspension In Chemistry The particles in suspensions are larger than those found in solutions. Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. — the suspension is a heterogeneous mixture in which the solute. Explore all the neet chemistry. solutions are typically clear and transparent due to. Opposite Of Suspension In Chemistry.
From www.youtube.com
What is Suspension and Properties of Suspension Surface Chemistry Opposite Of Suspension In Chemistry a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. — the suspension is a heterogeneous mixture in which the solute. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. Components of a suspension can. Opposite Of Suspension In Chemistry.
From www.youtube.com
Class 6 Science Chapter 7 Uses of solution, suspension uses properties Opposite Of Suspension In Chemistry — the table below shows the main differences between suspension and emulsion: a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. — the suspension is a heterogeneous mixture in which the solute. — a suspension is a mixture of particles with diameters of about 1 µm (1000. Opposite Of Suspension In Chemistry.
From exobkcoth.blob.core.windows.net
Definition Of Suspension In Chemistry Class 9 at Jeff Obryan blog Opposite Of Suspension In Chemistry Explore all the neet chemistry. — the suspension is a heterogeneous mixture in which the solute. solutions are typically clear and transparent due to the small size of the solute particles. The particles in suspensions are larger than those found in solutions. — the table below shows the main differences between suspension and emulsion: Components of a. Opposite Of Suspension In Chemistry.
From www.slideshare.net
Solutions, suspensions, and colloids Opposite Of Suspension In Chemistry — the suspension is a heterogeneous mixture in which the solute. The particles in suspensions are larger than those found in solutions. Explore all the neet chemistry. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. a colloid is a mixture in which. Opposite Of Suspension In Chemistry.
From dxorowdpy.blob.core.windows.net
Suspension Analytical Chemistry at Daniel Glover blog Opposite Of Suspension In Chemistry — the suspension is a heterogeneous mixture in which the solute. a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. — a suspension is a heterogeneous mixture in which particles disperse within a liquid or gas (fluid) but. — a suspension is a mixture of particles with. Opposite Of Suspension In Chemistry.
From exouqcyfe.blob.core.windows.net
What Is Colloidal Suspension In Chemistry at Robert Lemond blog Opposite Of Suspension In Chemistry This clarity allows light to pass through the solution without. a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. — the table below shows the main differences between suspension and emulsion: Explore all the neet chemistry. — a suspension is a mixture of particles with diameters of about. Opposite Of Suspension In Chemistry.
From slidetodoc.com
UNDERSTANDING SOLUTIONS PART 1 SOLUTIONS SUSPENSIONS A suspension Opposite Of Suspension In Chemistry Explore all the neet chemistry. — a suspension is a heterogeneous mixture in which particles disperse within a liquid or gas (fluid) but. This clarity allows light to pass through the solution without. solutions are typically clear and transparent due to the small size of the solute particles. — the table below shows the main differences between. Opposite Of Suspension In Chemistry.
From www.slideshare.net
Properties of suspensions Opposite Of Suspension In Chemistry Explore all the neet chemistry. — the suspension is a heterogeneous mixture in which the solute. This clarity allows light to pass through the solution without. — the table below shows the main differences between suspension and emulsion: a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. . Opposite Of Suspension In Chemistry.
From dxojgzbqt.blob.core.windows.net
Suspension In Chemistry at Kirkham blog Opposite Of Suspension In Chemistry Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. The particles in suspensions are larger than those found in solutions. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. —. Opposite Of Suspension In Chemistry.
From in.pinterest.com
Solutions, suspensions, and colloids Suspension mixture, Body diagram Opposite Of Suspension In Chemistry Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. — the suspension is a heterogeneous mixture in which the solute. This clarity allows light to pass through the solution without. — a suspension is a mixture of particles with diameters of about 1 µm. Opposite Of Suspension In Chemistry.
From www.youtube.com
Difference Between Solutions And Suspensions, General Science Lecture Opposite Of Suspension In Chemistry a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. The particles in suspensions are larger than those found in solutions. — a suspension is a. Opposite Of Suspension In Chemistry.
From www.youtube.com
Suspensions, colloids and solutions Chemistry Khan Academy YouTube Opposite Of Suspension In Chemistry — the suspension is a heterogeneous mixture in which the solute. This clarity allows light to pass through the solution without. Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. The particles in suspensions are larger than those found in solutions. Explore all the neet. Opposite Of Suspension In Chemistry.
From dxorvtvqp.blob.core.windows.net
Suspension In Chemistry Simple at Samuel Speed blog Opposite Of Suspension In Chemistry This clarity allows light to pass through the solution without. The particles in suspensions are larger than those found in solutions. — the table below shows the main differences between suspension and emulsion: — the suspension is a heterogeneous mixture in which the solute. solutions are typically clear and transparent due to the small size of the. Opposite Of Suspension In Chemistry.
From byjus.com
Suspensions (Chemistry) Definition, Properties, Examples with Videos Opposite Of Suspension In Chemistry This clarity allows light to pass through the solution without. solutions are typically clear and transparent due to the small size of the solute particles. — a suspension is a heterogeneous mixture in which particles disperse within a liquid or gas (fluid) but. Components of a suspension can be evenly distributed by mechanical means, like by shaking the. Opposite Of Suspension In Chemistry.
From byjus.com
Suspensions (Chemistry) Definition, Properties, Examples with Videos Opposite Of Suspension In Chemistry a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. The particles in suspensions are larger than those found in solutions. This clarity allows light to pass through. Opposite Of Suspension In Chemistry.
From dxojgzbqt.blob.core.windows.net
Suspension In Chemistry at Kirkham blog Opposite Of Suspension In Chemistry — the suspension is a heterogeneous mixture in which the solute. Explore all the neet chemistry. The particles in suspensions are larger than those found in solutions. — a suspension is a heterogeneous mixture in which particles disperse within a liquid or gas (fluid) but. — the table below shows the main differences between suspension and emulsion:. Opposite Of Suspension In Chemistry.
From brainly.in
How are sol, solution and suspension different from each other Opposite Of Suspension In Chemistry — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. solutions are typically clear and transparent due to the small size of the solute particles. Explore all the neet chemistry. This clarity allows light to pass through the solution without. The particles in suspensions are. Opposite Of Suspension In Chemistry.
From www.pinterest.com
Suspension Easy Science Chemistry study guide, Physics concepts Opposite Of Suspension In Chemistry a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. — the suspension is a heterogeneous mixture in which the solute. The particles in suspensions are. Opposite Of Suspension In Chemistry.
From dxojgzbqt.blob.core.windows.net
Suspension In Chemistry at Kirkham blog Opposite Of Suspension In Chemistry Explore all the neet chemistry. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. — the table below shows the main differences between suspension and emulsion: Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the. Opposite Of Suspension In Chemistry.
From www.vecteezy.com
Different dispersion system, true and colloidal solution and suspension Opposite Of Suspension In Chemistry — the table below shows the main differences between suspension and emulsion: a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. This clarity allows light to pass through the solution without. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that. Opposite Of Suspension In Chemistry.
From www.slideserve.com
PPT Chemistry PowerPoint Presentation, free download ID1073031 Opposite Of Suspension In Chemistry The particles in suspensions are larger than those found in solutions. This clarity allows light to pass through the solution without. Explore all the neet chemistry. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. — the table below shows the main differences between. Opposite Of Suspension In Chemistry.
From www.slideserve.com
PPT Solutions PowerPoint Presentation, free download ID5813207 Opposite Of Suspension In Chemistry Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. solutions are typically clear and transparent due to the small size of the solute particles. — the suspension is a heterogeneous mixture in which the solute. — a suspension is a mixture of particles. Opposite Of Suspension In Chemistry.
From dxojgzbqt.blob.core.windows.net
Suspension In Chemistry at Kirkham blog Opposite Of Suspension In Chemistry Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. — the suspension is a heterogeneous mixture in which the solute. — the table below shows. Opposite Of Suspension In Chemistry.
From kunduz.com
Homogeneous Mixtures Definition, Properties, Types, Examples Opposite Of Suspension In Chemistry a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. solutions are typically clear and transparent due to the small size of the solute particles. —. Opposite Of Suspension In Chemistry.
From www.slideserve.com
PPT Chapter 12 SOLUTIONS PowerPoint Presentation, free download ID Opposite Of Suspension In Chemistry — the table below shows the main differences between suspension and emulsion: This clarity allows light to pass through the solution without. The particles in suspensions are larger than those found in solutions. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. Components of. Opposite Of Suspension In Chemistry.
From www.majordifferences.com
Difference Between True Solutions, Colloidal solution and Suspension Opposite Of Suspension In Chemistry — the table below shows the main differences between suspension and emulsion: a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. Explore all the neet chemistry. The particles in suspensions are larger than those found in solutions. — a suspension is a mixture of particles with diameters of. Opposite Of Suspension In Chemistry.
From www.vrogue.co
Examples Of Chemical Suspensions vrogue.co Opposite Of Suspension In Chemistry Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. — the suspension is a heterogeneous mixture in which the solute. This clarity allows light to pass through the solution without. — a suspension is a heterogeneous mixture in which particles disperse within a liquid. Opposite Of Suspension In Chemistry.
From quizizz.com
Solution, Colloid and Suspension Science Quizizz Opposite Of Suspension In Chemistry — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. — the table below shows the main differences between suspension and emulsion:. Opposite Of Suspension In Chemistry.
From www.slideserve.com
PPT Suspension PowerPoint Presentation, free download ID4164419 Opposite Of Suspension In Chemistry This clarity allows light to pass through the solution without. — the table below shows the main differences between suspension and emulsion: — a suspension is a heterogeneous mixture in which particles disperse within a liquid or gas (fluid) but. The particles in suspensions are larger than those found in solutions. a colloid is a mixture in. Opposite Of Suspension In Chemistry.
From mungfali.com
5 Examples Of Suspension Opposite Of Suspension In Chemistry — the suspension is a heterogeneous mixture in which the solute. The particles in suspensions are larger than those found in solutions. Components of a suspension can be evenly distributed by mechanical means, like by shaking the contents but the components will eventually settle out. — a suspension is a heterogeneous mixture in which particles disperse within a. Opposite Of Suspension In Chemistry.
From www.slideshare.net
Ch08 solutions & suspensions Opposite Of Suspension In Chemistry — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. The particles in suspensions are larger than those found in solutions. a colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout. — the suspension is a. Opposite Of Suspension In Chemistry.
From www.youtube.com
Suspension Vs Syrup Definition Difference drhidayatpharmacist Opposite Of Suspension In Chemistry — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. — the table below shows the main differences between suspension and emulsion: — the suspension is a heterogeneous mixture in which the solute. solutions are typically clear and transparent due to the small. Opposite Of Suspension In Chemistry.
From exoqlkifs.blob.core.windows.net
Example Of Suspension at Susan Johnson blog Opposite Of Suspension In Chemistry The particles in suspensions are larger than those found in solutions. Explore all the neet chemistry. — the suspension is a heterogeneous mixture in which the solute. — a suspension is a mixture of particles with diameters of about 1 µm (1000 nm) that are distributed throughout a second phase. a colloid is a mixture in which. Opposite Of Suspension In Chemistry.