Data Cleaning Hippocampus
Ann Wells
April 23, 2023
tdata.FPKM.sample.info <- readRDS(here("Data","20190406_RNAseq_B6_4wk_2DG_counts_phenotypes.RData"))
tdata.FPKM <- readRDS(here("Data","20190406_RNAseq_B6_4wk_2DG_counts_numeric.RData"))
log.tdata.FPKM <- log(tdata.FPKM + 1)
log.tdata.FPKM <- as.data.frame(log.tdata.FPKM)
log.tdata.FPKM.sample.info <- cbind(log.tdata.FPKM, tdata.FPKM.sample.info[,27238:27240])
log.tdata.FPKM.sample.info <- log.tdata.FPKM.sample.info %>% rownames_to_column() %>% filter(rowname != "A113") %>% column_to_rownames()
log.tdata.FPKM.subset <- log.tdata.FPKM[,colMeans(log.tdata.FPKM != 0) > 0.5]
log.tdata.FPKM.sample.info.subset <- cbind(log.tdata.FPKM.subset,tdata.FPKM.sample.info[,27238:27240])
log.tdata.FPKM.sample.info.subset <- log.tdata.FPKM.sample.info.subset %>% rownames_to_column() %>% filter(rowname != "A113") %>% column_to_rownames()
log.tdata.FPKM.sample.info.subset.hippocampus <- log.tdata.FPKM.sample.info.subset %>% rownames_to_column() %>% filter(Tissue == "Hippocampus") %>% column_to_rownames()Wrangle Data
I will use Mouse IDs, Treatment, and Time to keep track of the values in the matrices. All other covariates will be discarded.
# Set rownames by mouse ID and tissue
rownames(log.tdata.FPKM.sample.info.subset.hippocampus) <- paste0(rownames(log.tdata.FPKM.sample.info.subset.hippocampus),":", log.tdata.FPKM.sample.info.subset.hippocampus$Time, ":", log.tdata.FPKM.sample.info.subset.hippocampus$Treatment)
# Discard covariates from columns 17333-17336
log.tdata.FPKM.sample.info.subset.hippocampus <- log.tdata.FPKM.sample.info.subset.hippocampus[,-(17333:17336)]
head(log.tdata.FPKM.sample.info.subset.hippocampus[,1:5])## ENSMUSG00000000001 ENSMUSG00000000028 ENSMUSG00000000031
## A008:4 wks:None 2.889816 0.7129498 0.2776317
## A017:96 hrs:2DG 2.731115 1.1847900 0.1133287
## A026:4 wks:2DG 2.915606 1.1662709 0.1397619
## A035:4 wks:2DG 2.683758 0.5128236 0.1222176
## A044:4 wks:None 2.933325 0.8329091 0.2070142
## A053:4 wks:None 2.584752 0.5877867 0.2926696
## ENSMUSG00000000037 ENSMUSG00000000049
## A008:4 wks:None 0.3148107 0.6471032
## A017:96 hrs:2DG 0.2468601 0.3364722
## A026:4 wks:2DG 0.6780335 0.8109302
## A035:4 wks:2DG 0.5364934 0.3646431
## A044:4 wks:None 0.3506569 0.0000000
## A053:4 wks:None 0.6151856 0.2926696Check Data for Missing Values
WGCNA will have poor results if the data have too many missing values. I checked if any metabolites fall into this category.
log.tdata.FPKM.sample.info.subset.hippocampus.missing <- missing(log.tdata.FPKM.sample.info.subset.hippocampus)
cat("logFPKM: ", goodSamplesGenes(log.tdata.FPKM.sample.info.subset.hippocampus.missing, verbose=0)$allOK, "\n")## logFPKM: TRUEWGCNA reports that all data are good! I now use hierarchical clustering to detect any obvious outliers. I did not see any particularly egregious outliers.
sampleclustering(log.tdata.FPKM.sample.info.subset.hippocampus.missing)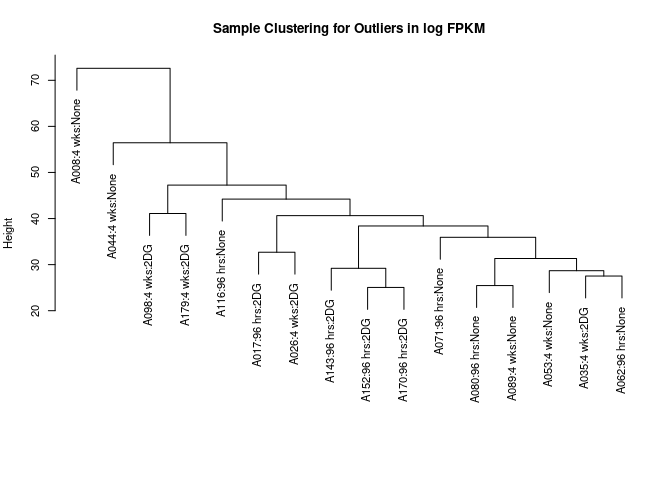
saveRDS(log.tdata.FPKM.sample.info.subset.hippocampus.missing, here("Data","Hippocampus","log.tdata.FPKM.sample.info.subset.hippocampus.missing.WGCNA.RData"))Analysis performed by Ann Wells
The Carter Lab The Jackson Laboratory 2023
ann.wells@jax.org